| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 205224 | 461101 | 2016 | 7 صفحه PDF | دانلود رایگان |
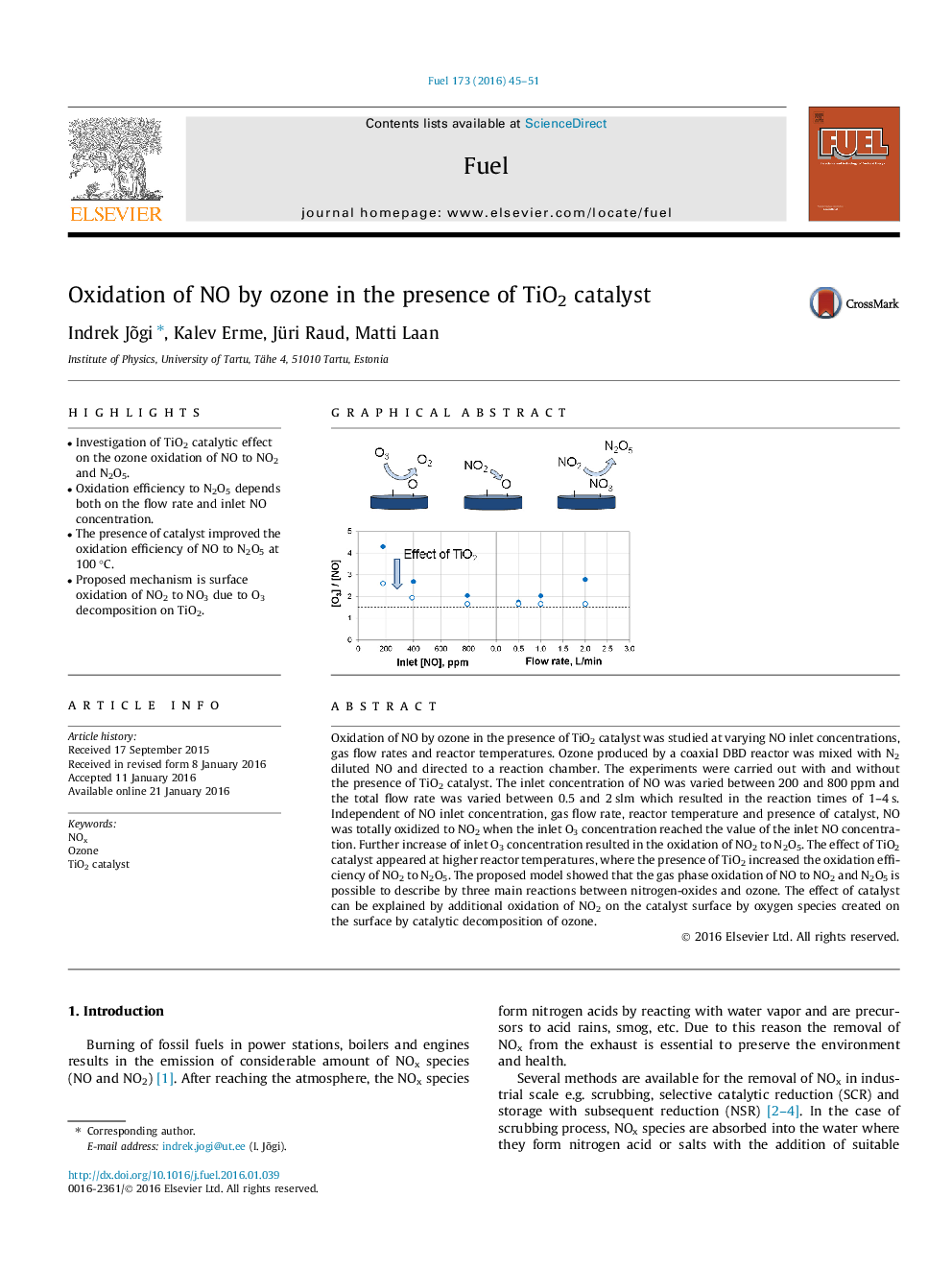
• Investigation of TiO2 catalytic effect on the ozone oxidation of NO to NO2 and N2O5.
• Oxidation efficiency to N2O5 depends both on the flow rate and inlet NO concentration.
• The presence of catalyst improved the oxidation efficiency of NO to N2O5 at 100 °C.
• Proposed mechanism is surface oxidation of NO2 to NO3 due to O3 decomposition on TiO2.
Oxidation of NO by ozone in the presence of TiO2 catalyst was studied at varying NO inlet concentrations, gas flow rates and reactor temperatures. Ozone produced by a coaxial DBD reactor was mixed with N2 diluted NO and directed to a reaction chamber. The experiments were carried out with and without the presence of TiO2 catalyst. The inlet concentration of NO was varied between 200 and 800 ppm and the total flow rate was varied between 0.5 and 2 slm which resulted in the reaction times of 1–4 s. Independent of NO inlet concentration, gas flow rate, reactor temperature and presence of catalyst, NO was totally oxidized to NO2 when the inlet O3 concentration reached the value of the inlet NO concentration. Further increase of inlet O3 concentration resulted in the oxidation of NO2 to N2O5. The effect of TiO2 catalyst appeared at higher reactor temperatures, where the presence of TiO2 increased the oxidation efficiency of NO2 to N2O5. The proposed model showed that the gas phase oxidation of NO to NO2 and N2O5 is possible to describe by three main reactions between nitrogen-oxides and ozone. The effect of catalyst can be explained by additional oxidation of NO2 on the catalyst surface by oxygen species created on the surface by catalytic decomposition of ozone.
Figure optionsDownload as PowerPoint slide
Journal: Fuel - Volume 173, 1 June 2016, Pages 45–51