| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1229003 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2015 | 15 Pages |
•A novel bidentate NO Schiff base ligand was synthesized and characterized.•The Schiff base ligand was allowed to react with a variety of transition metal ions.•Binuclear complexes were synthesized and characterized.•The ligand and its metal complexes showed variable antimicrobial activity.•Molecular docking of the compounds showed various interactions with the active sites of dihydrofolate reductase enzyme.
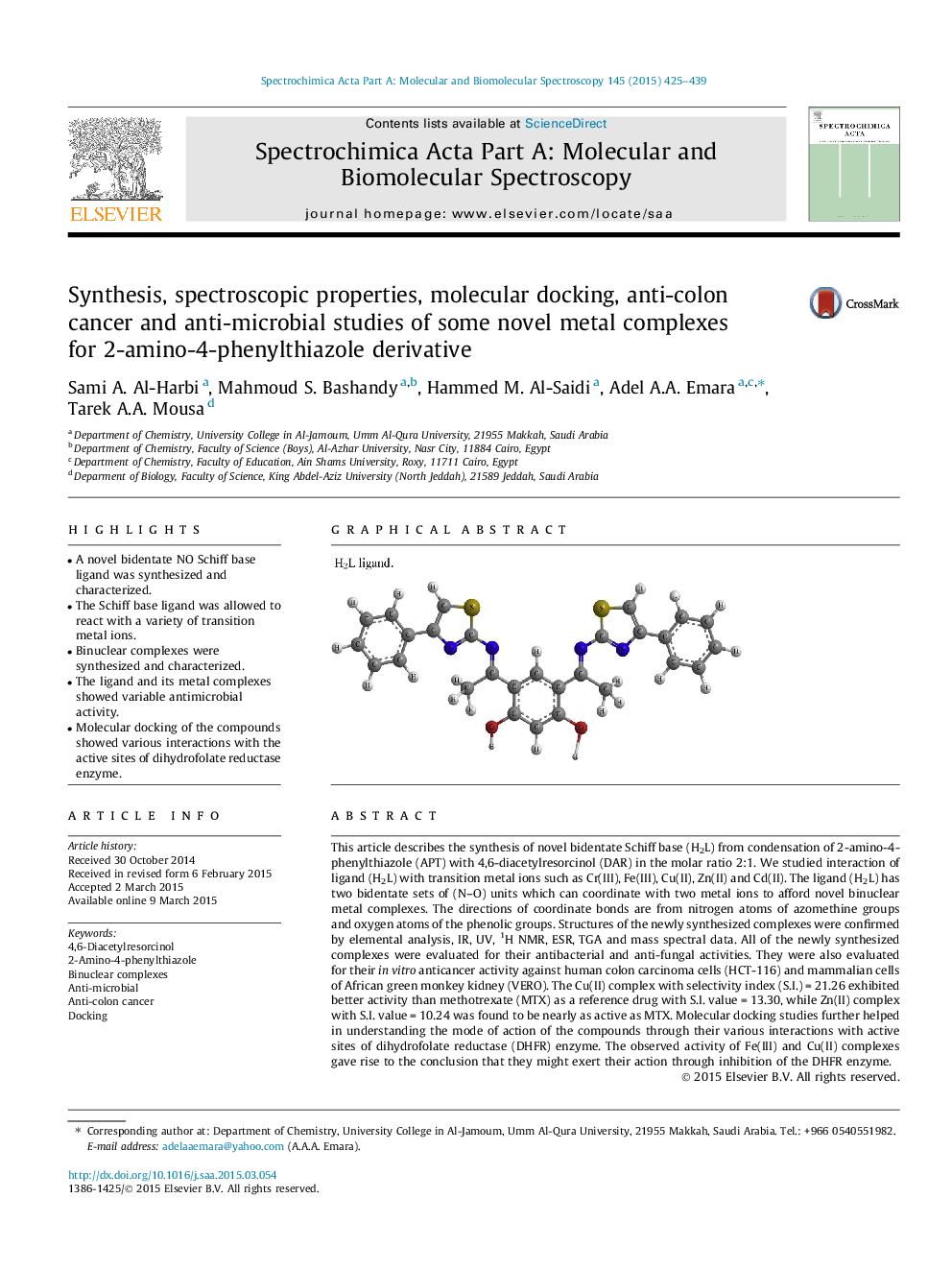
This article describes the synthesis of novel bidentate Schiff base (H2L) from condensation of 2-amino-4-phenylthiazole (APT) with 4,6-diacetylresorcinol (DAR) in the molar ratio 2:1. We studied interaction of ligand (H2L) with transition metal ions such as Cr(III), Fe(III), Cu(II), Zn(II) and Cd(II). The ligand (H2L) has two bidentate sets of (N–O) units which can coordinate with two metal ions to afford novel binuclear metal complexes. The directions of coordinate bonds are from nitrogen atoms of azomethine groups and oxygen atoms of the phenolic groups. Structures of the newly synthesized complexes were confirmed by elemental analysis, IR, UV, 1H NMR, ESR, TGA and mass spectral data. All of the newly synthesized complexes were evaluated for their antibacterial and anti-fungal activities. They were also evaluated for their in vitro anticancer activity against human colon carcinoma cells (HCT-116) and mammalian cells of African green monkey kidney (VERO). The Cu(II) complex with selectivity index (S.I.) = 21.26 exhibited better activity than methotrexate (MTX) as a reference drug with S.I. value = 13.30, while Zn(II) complex with S.I. value = 10.24 was found to be nearly as active as MTX. Molecular docking studies further helped in understanding the mode of action of the compounds through their various interactions with active sites of dihydrofolate reductase (DHFR) enzyme. The observed activity of Fe(III) and Cu(II) complexes gave rise to the conclusion that they might exert their action through inhibition of the DHFR enzyme.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide