| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1229646 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2014 | 9 Pages |
•The interactions between BSA and FNC or analogs have been investigated.•Hydrophobic interactions play major role in the binding process.•The influence of molecular structure on the binding aspects has been investigated.•Molecular docking was also applied in the binding study.
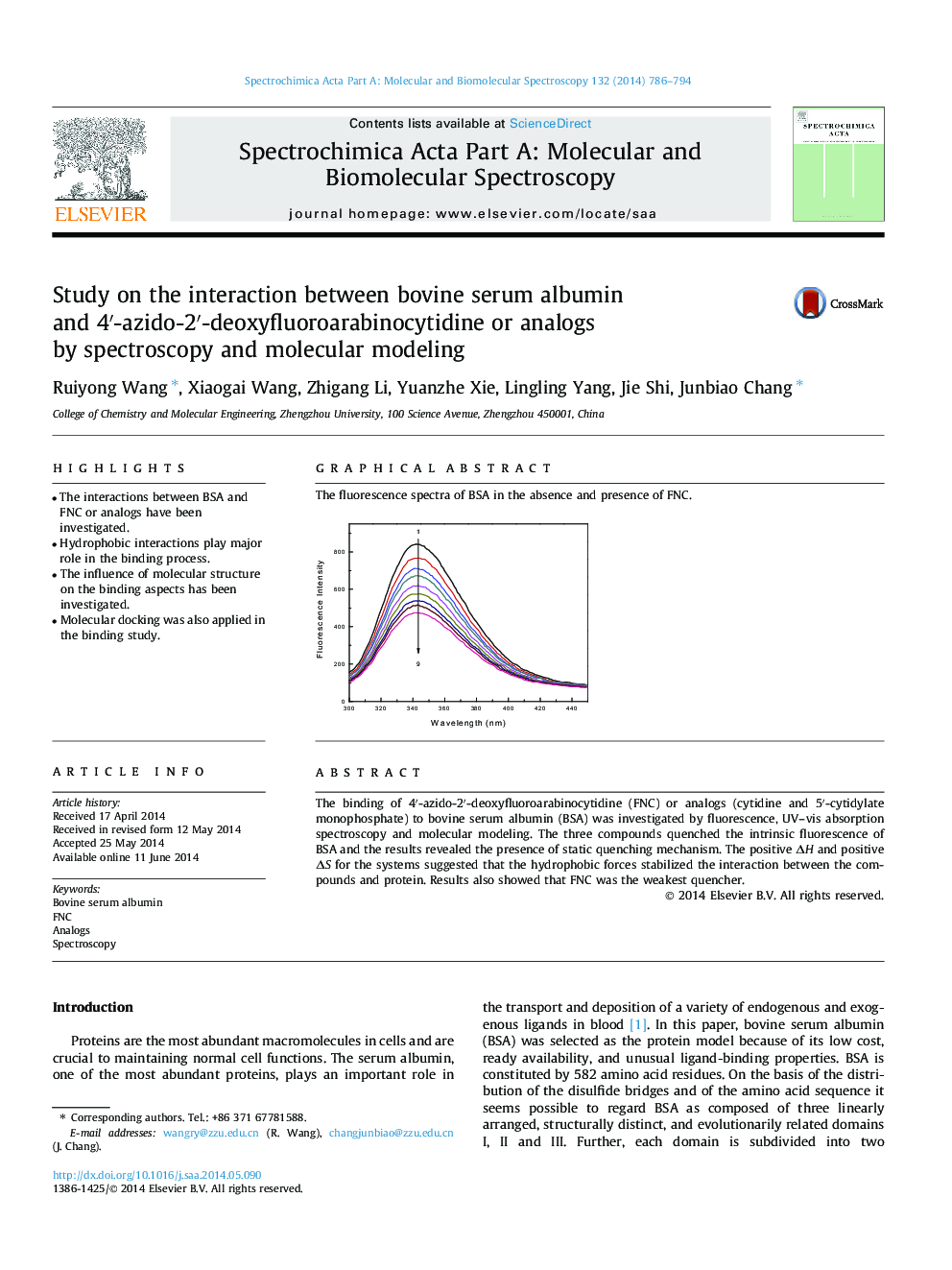
The binding of 4′-azido-2′-deoxyfluoroarabinocytidine (FNC) or analogs (cytidine and 5′-cytidylate monophosphate) to bovine serum albumin (BSA) was investigated by fluorescence, UV–vis absorption spectroscopy and molecular modeling. The three compounds quenched the intrinsic fluorescence of BSA and the results revealed the presence of static quenching mechanism. The positive ΔH and positive ΔS for the systems suggested that the hydrophobic forces stabilized the interaction between the compounds and protein. Results also showed that FNC was the weakest quencher.
Graphical abstractThe fluorescence spectra of BSA in the absence and presence of FNC.Figure optionsDownload full-size imageDownload as PowerPoint slide