| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1229667 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2015 | 9 Pages |
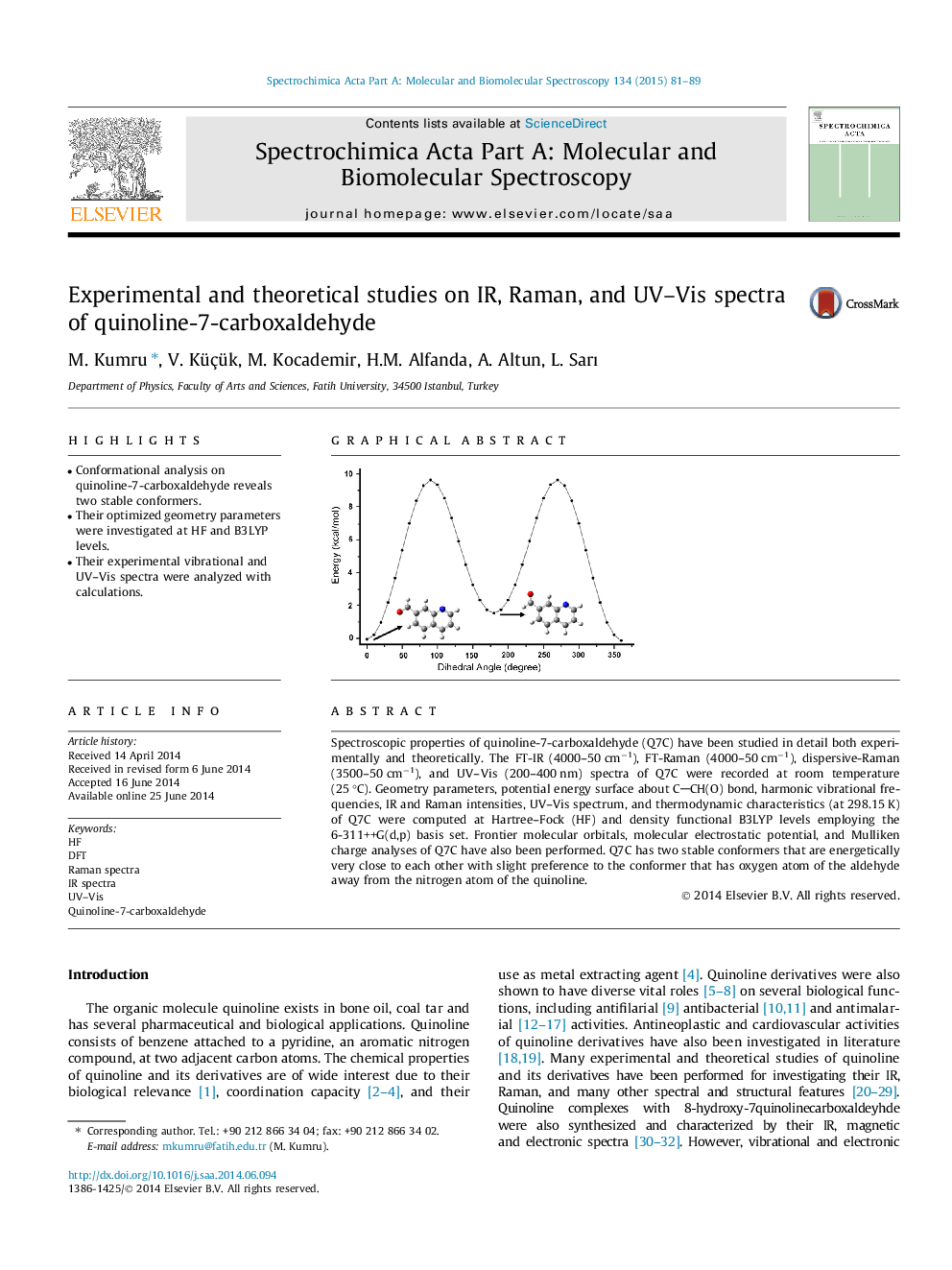
•Conformational analysis on quinoline-7-carboxaldehyde reveals two stable conformers.•Their optimized geometry parameters were investigated at HF and B3LYP levels.•Their experimental vibrational and UV–Vis spectra were analyzed with calculations.
Spectroscopic properties of quinoline-7-carboxaldehyde (Q7C) have been studied in detail both experimentally and theoretically. The FT-IR (4000–50 cm−1), FT-Raman (4000–50 cm−1), dispersive-Raman (3500–50 cm−1), and UV–Vis (200–400 nm) spectra of Q7C were recorded at room temperature (25 °C). Geometry parameters, potential energy surface about CCH(O) bond, harmonic vibrational frequencies, IR and Raman intensities, UV–Vis spectrum, and thermodynamic characteristics (at 298.15 K) of Q7C were computed at Hartree–Fock (HF) and density functional B3LYP levels employing the 6-311++G(d,p) basis set. Frontier molecular orbitals, molecular electrostatic potential, and Mulliken charge analyses of Q7C have also been performed. Q7C has two stable conformers that are energetically very close to each other with slight preference to the conformer that has oxygen atom of the aldehyde away from the nitrogen atom of the quinoline.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide