| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1230090 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2015 | 7 Pages |
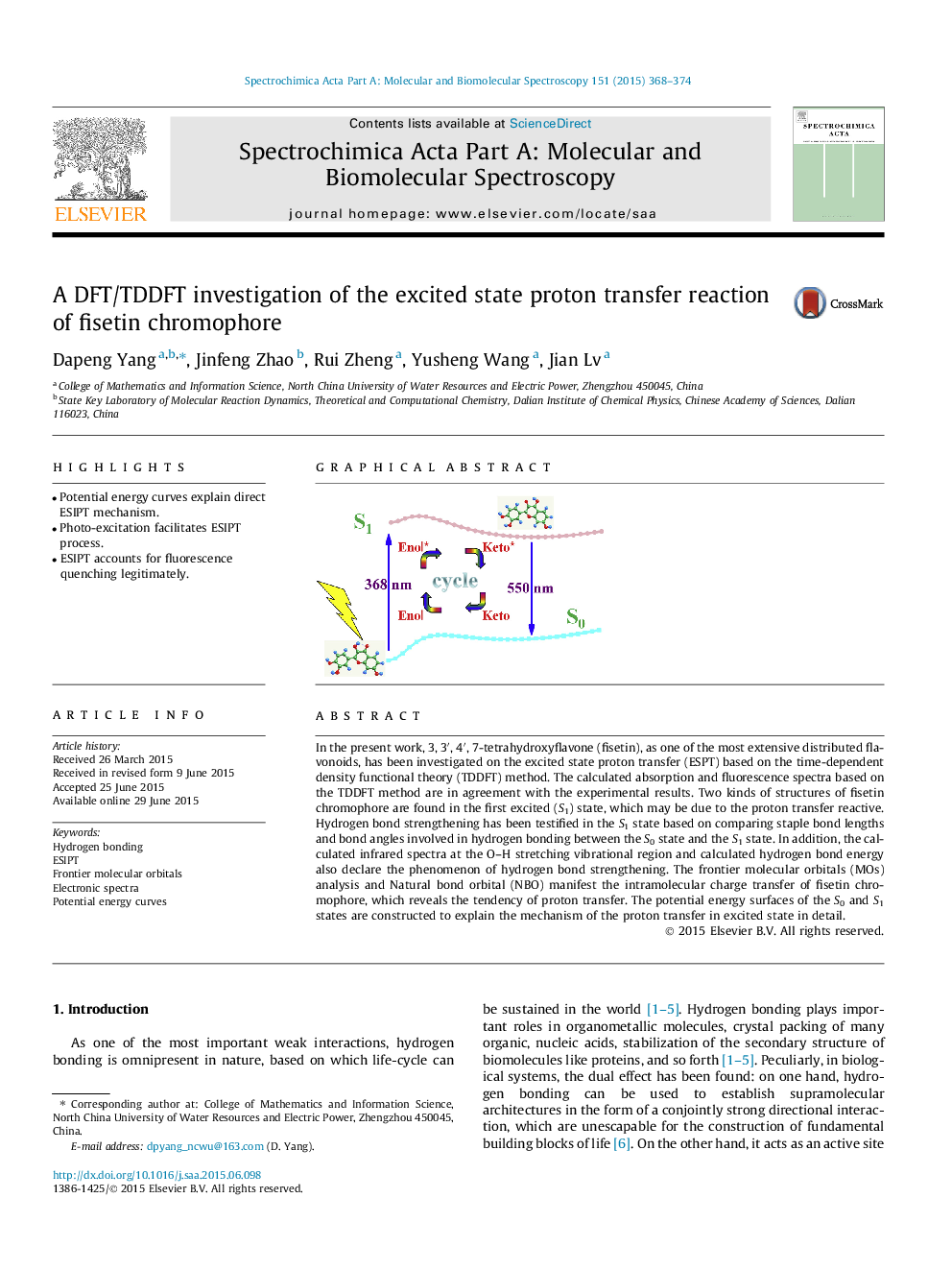
•Potential energy curves explain direct ESIPT mechanism.•Photo-excitation facilitates ESIPT process.•ESIPT accounts for fluorescence quenching legitimately.
In the present work, 3, 3′, 4′, 7-tetrahydroxyflavone (fisetin), as one of the most extensive distributed flavonoids, has been investigated on the excited state proton transfer (ESPT) based on the time-dependent density functional theory (TDDFT) method. The calculated absorption and fluorescence spectra based on the TDDFT method are in agreement with the experimental results. Two kinds of structures of fisetin chromophore are found in the first excited (S1) state, which may be due to the proton transfer reactive. Hydrogen bond strengthening has been testified in the S1 state based on comparing staple bond lengths and bond angles involved in hydrogen bonding between the S0 state and the S1 state. In addition, the calculated infrared spectra at the O–H stretching vibrational region and calculated hydrogen bond energy also declare the phenomenon of hydrogen bond strengthening. The frontier molecular orbitals (MOs) analysis and Natural bond orbital (NBO) manifest the intramolecular charge transfer of fisetin chromophore, which reveals the tendency of proton transfer. The potential energy surfaces of the S0 and S1 states are constructed to explain the mechanism of the proton transfer in excited state in detail.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide