| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1230125 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2015 | 7 Pages |
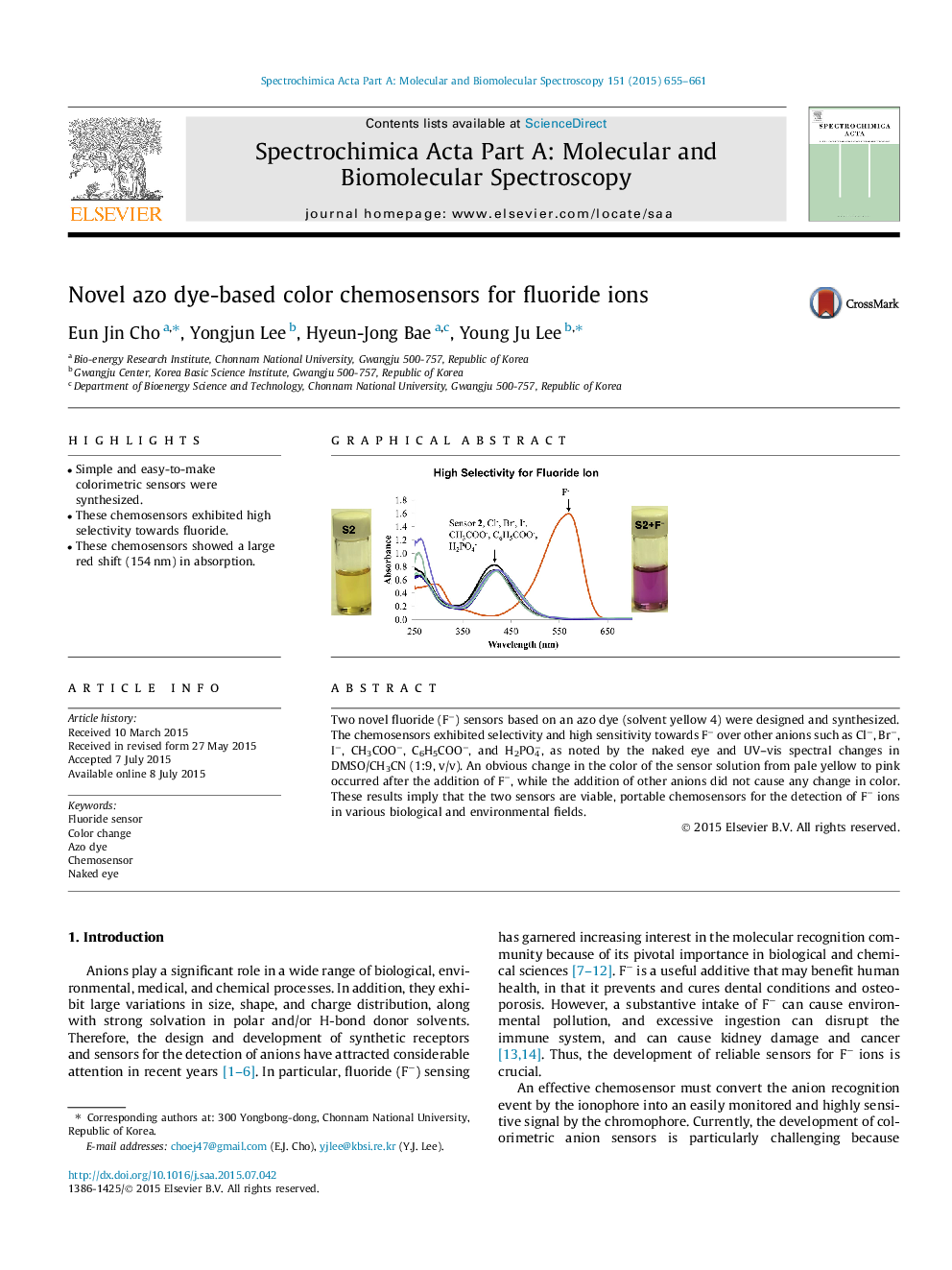
•Simple and easy-to-make colorimetric sensors were synthesized.•These chemosensors exhibited high selectivity towards fluoride.•These chemosensors showed a large red shift (154 nm) in absorption.
Two novel fluoride (F−) sensors based on an azo dye (solvent yellow 4) were designed and synthesized. The chemosensors exhibited selectivity and high sensitivity towards F− over other anions such as Cl−, Br−, I−, CH3COO−, C6H5COO−, and H2PO4−, as noted by the naked eye and UV–vis spectral changes in DMSO/CH3CN (1:9, v/v). An obvious change in the color of the sensor solution from pale yellow to pink occurred after the addition of F−, while the addition of other anions did not cause any change in color. These results imply that the two sensors are viable, portable chemosensors for the detection of F− ions in various biological and environmental fields.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide