| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1230296 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2017 | 9 Pages |
Abstract
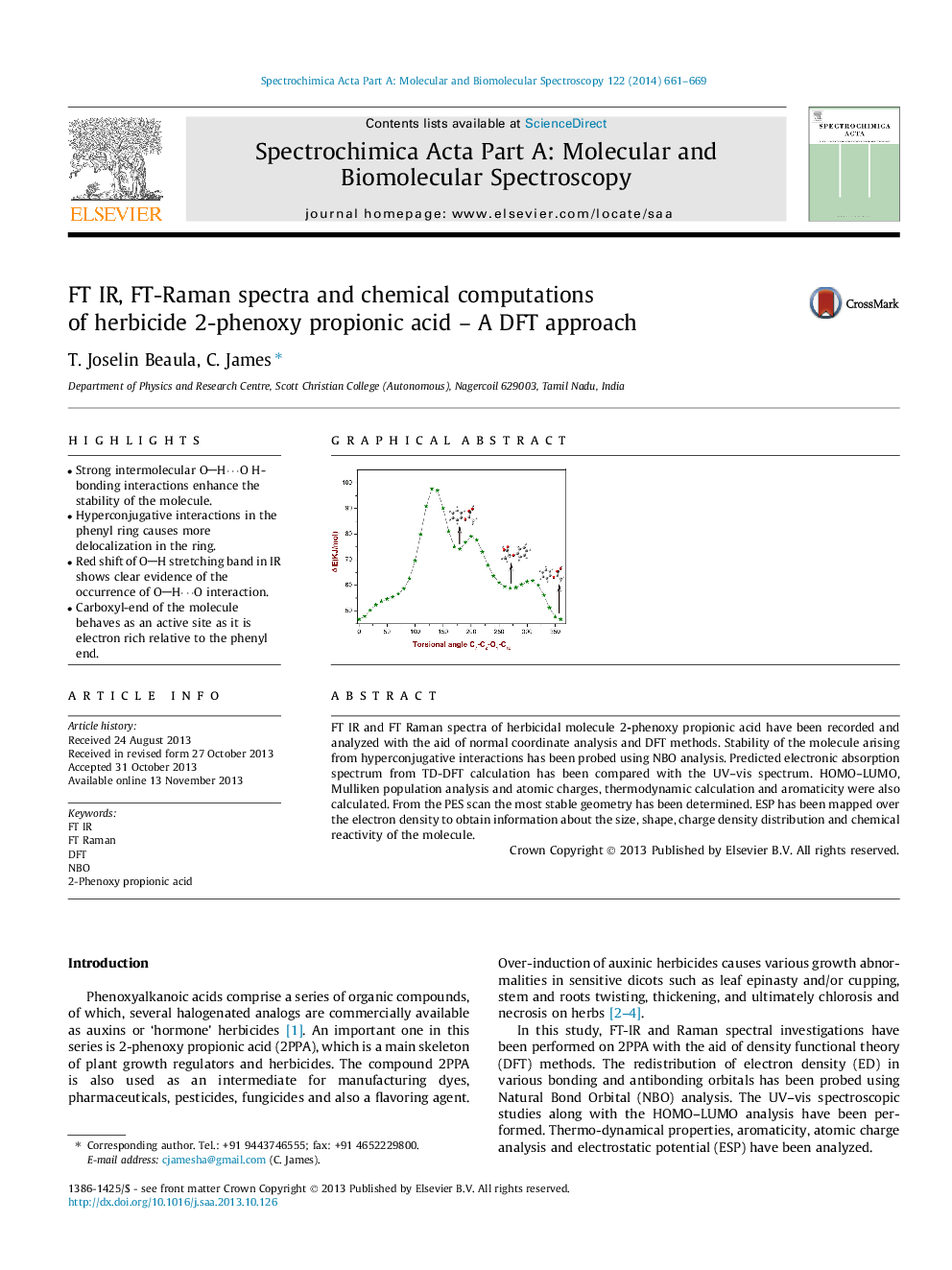
- Strong intermolecular OHâ¯O H-bonding interactions enhance the stability of the molecule.
- Hyperconjugative interactions in the phenyl ring causes more delocalization in the ring.
- Red shift of OH stretching band in IR shows clear evidence of the occurrence of OHâ¯O interaction.
- Carboxyl-end of the molecule behaves as an active site as it is electron rich relative to the phenyl end.
Related Topics
Physical Sciences and Engineering
Chemistry
Analytical Chemistry
Authors
T. Joselin Beaula, C. James,