| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1231531 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2013 | 5 Pages |
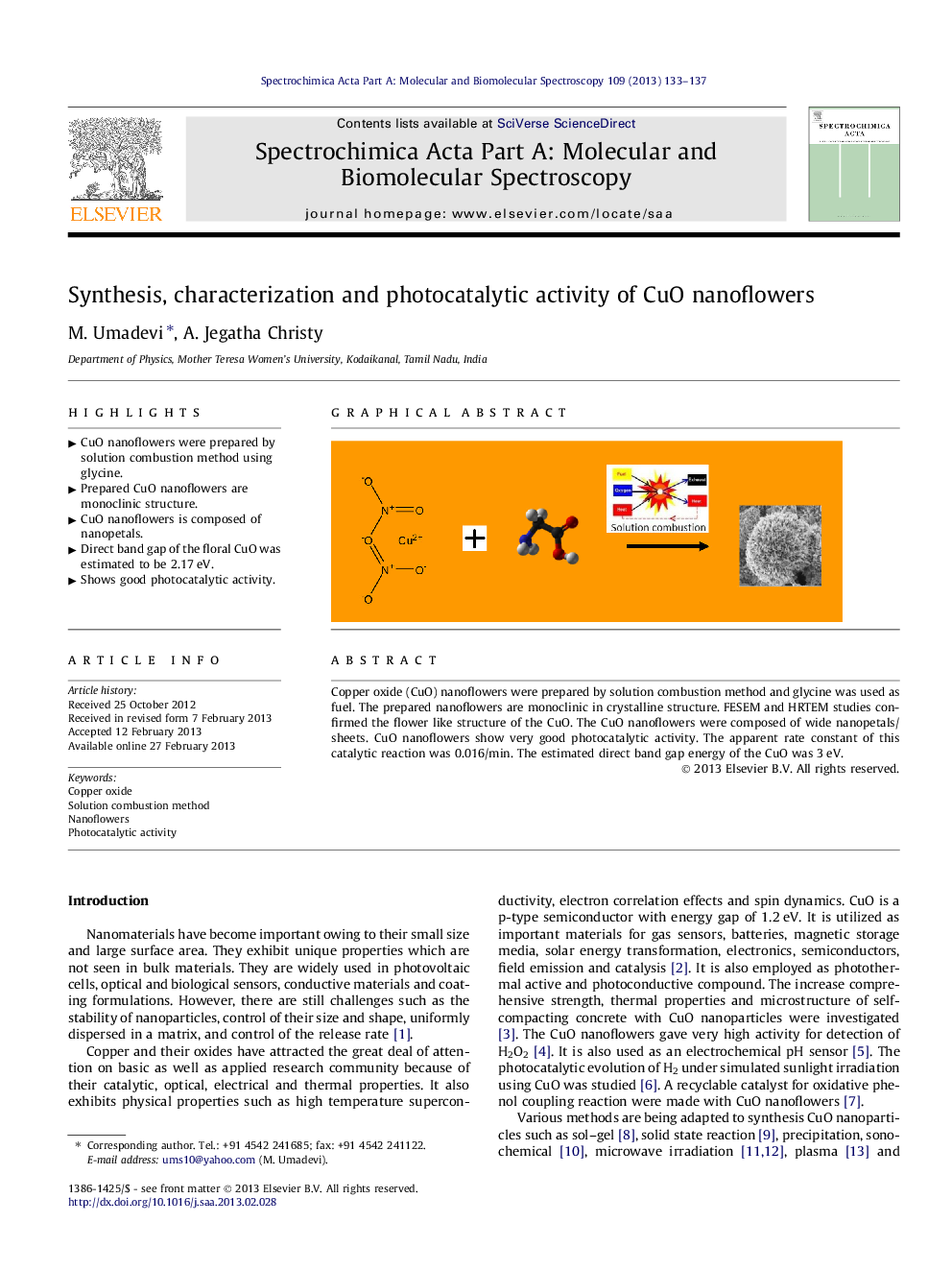
Copper oxide (CuO) nanoflowers were prepared by solution combustion method and glycine was used as fuel. The prepared nanoflowers are monoclinic in crystalline structure. FESEM and HRTEM studies confirmed the flower like structure of the CuO. The CuO nanoflowers were composed of wide nanopetals/sheets. CuO nanoflowers show very good photocatalytic activity. The apparent rate constant of this catalytic reaction was 0.016/min. The estimated direct band gap energy of the CuO was 3 eV.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slideHighlights► CuO nanoflowers were prepared by solution combustion method using glycine. ► Prepared CuO nanoflowers are monoclinic structure. ► CuO nanoflowers is composed of nanopetals. ► Direct band gap of the floral CuO was estimated to be 2.17 eV. ► Shows good photocatalytic activity.