| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1232507 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2015 | 6 Pages |
•7-Amino-4-methylcoumarin has been quenched by all TEMPO derivatives.•The fluorescence quenching mechanism was proved to be totally dynamic.•It seems that the fluorescence quenching is diffusion-limited caused by nonradiative processes.•Bimolecular quenching constants were determined.
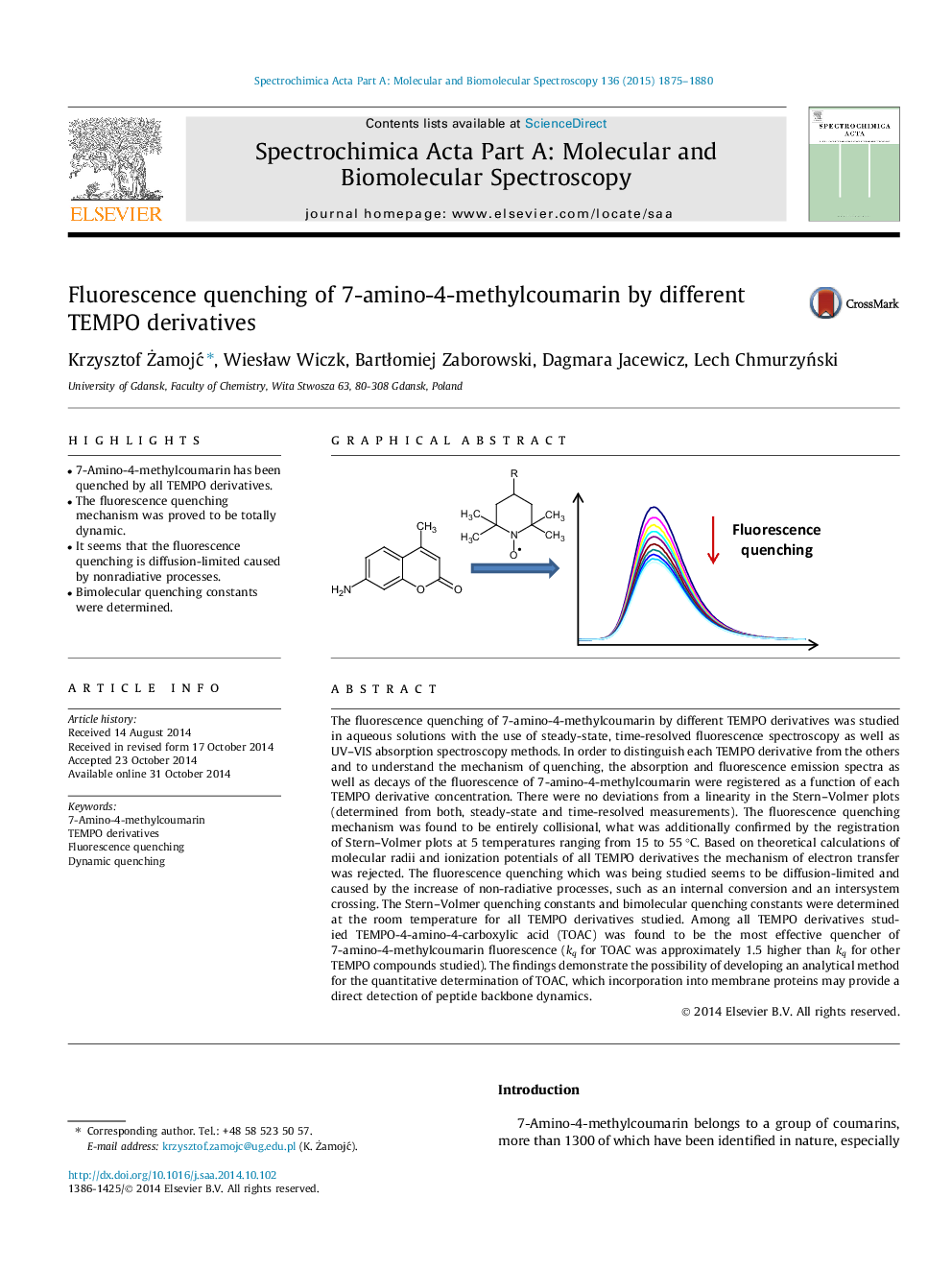
The fluorescence quenching of 7-amino-4-methylcoumarin by different TEMPO derivatives was studied in aqueous solutions with the use of steady-state, time-resolved fluorescence spectroscopy as well as UV–VIS absorption spectroscopy methods. In order to distinguish each TEMPO derivative from the others and to understand the mechanism of quenching, the absorption and fluorescence emission spectra as well as decays of the fluorescence of 7-amino-4-methylcoumarin were registered as a function of each TEMPO derivative concentration. There were no deviations from a linearity in the Stern–Volmer plots (determined from both, steady-state and time-resolved measurements). The fluorescence quenching mechanism was found to be entirely collisional, what was additionally confirmed by the registration of Stern–Volmer plots at 5 temperatures ranging from 15 to 55 °C. Based on theoretical calculations of molecular radii and ionization potentials of all TEMPO derivatives the mechanism of electron transfer was rejected. The fluorescence quenching which was being studied seems to be diffusion-limited and caused by the increase of non-radiative processes, such as an internal conversion and an intersystem crossing. The Stern–Volmer quenching constants and bimolecular quenching constants were determined at the room temperature for all TEMPO derivatives studied. Among all TEMPO derivatives studied TEMPO-4-amino-4-carboxylic acid (TOAC) was found to be the most effective quencher of 7-amino-4-methylcoumarin fluorescence (kq for TOAC was approximately 1.5 higher than kq for other TEMPO compounds studied). The findings demonstrate the possibility of developing an analytical method for the quantitative determination of TOAC, which incorporation into membrane proteins may provide a direct detection of peptide backbone dynamics.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide