| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1233422 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2014 | 6 Pages |
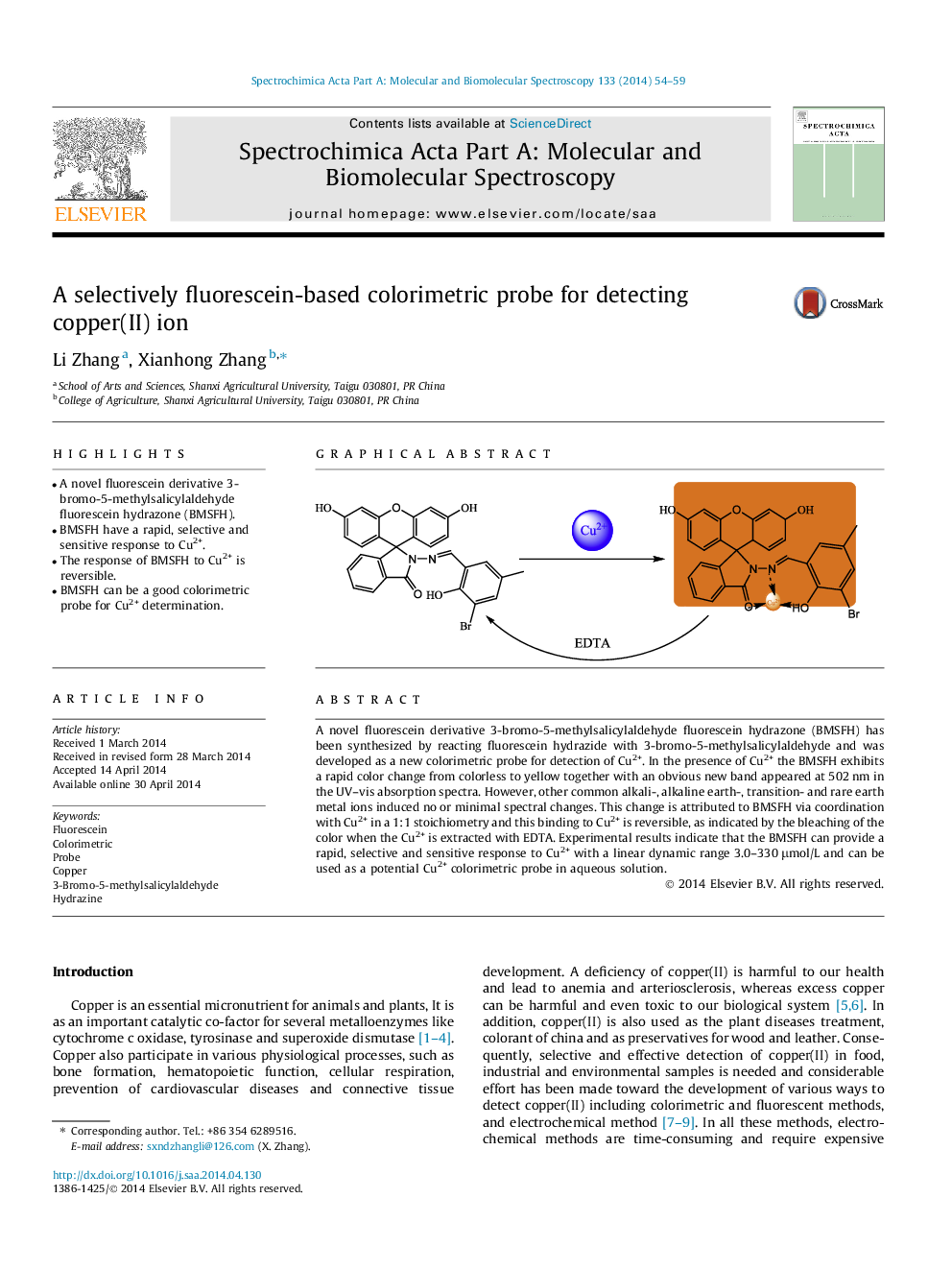
•A novel fluorescein derivative 3-bromo-5-methylsalicylaldehyde fluorescein hydrazone (BMSFH).•BMSFH have a rapid, selective and sensitive response to Cu2+.•The response of BMSFH to Cu2+ is reversible.•BMSFH can be a good colorimetric probe for Cu2+ determination.
A novel fluorescein derivative 3-bromo-5-methylsalicylaldehyde fluorescein hydrazone (BMSFH) has been synthesized by reacting fluorescein hydrazide with 3-bromo-5-methylsalicylaldehyde and was developed as a new colorimetric probe for detection of Cu2+. In the presence of Cu2+ the BMSFH exhibits a rapid color change from colorless to yellow together with an obvious new band appeared at 502 nm in the UV–vis absorption spectra. However, other common alkali-, alkaline earth-, transition- and rare earth metal ions induced no or minimal spectral changes. This change is attributed to BMSFH via coordination with Cu2+ in a 1:1 stoichiometry and this binding to Cu2+ is reversible, as indicated by the bleaching of the color when the Cu2+ is extracted with EDTA. Experimental results indicate that the BMSFH can provide a rapid, selective and sensitive response to Cu2+ with a linear dynamic range 3.0–330 μmol/L and can be used as a potential Cu2+ colorimetric probe in aqueous solution.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide