| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1233469 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2014 | 7 Pages |
•Iron(III)–oxazoline complex is applied in oxidation of sulfides.•Urea hydrogen peroxide (UHP) as oxidant.•Excellent yield and selectivity has been achieved.
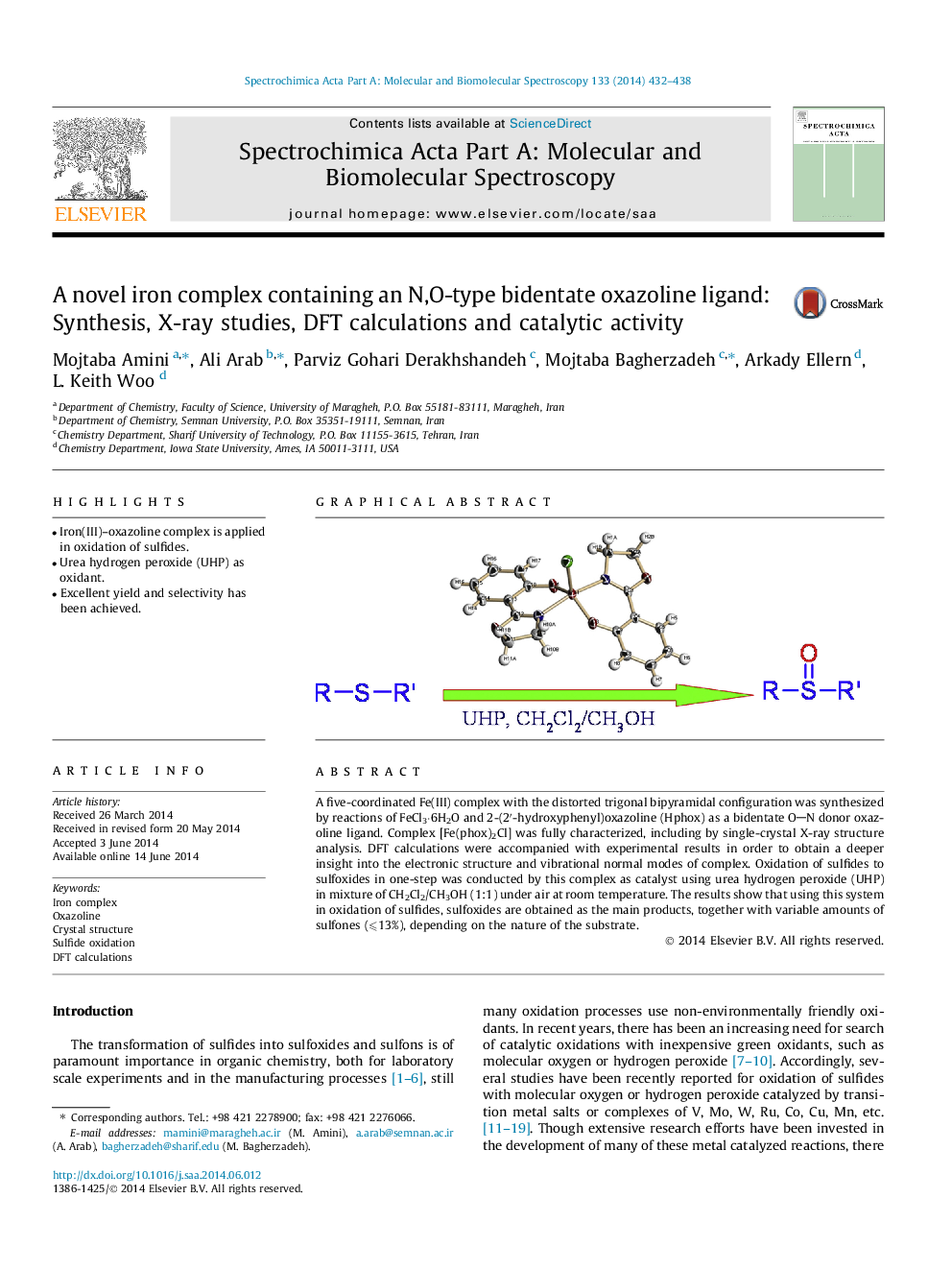
A five-coordinated Fe(III) complex with the distorted trigonal bipyramidal configuration was synthesized by reactions of FeCl3⋅6H2O and 2-(2′-hydroxyphenyl)oxazoline (Hphox) as a bidentate ON donor oxazoline ligand. Complex [Fe(phox)2Cl] was fully characterized, including by single-crystal X-ray structure analysis. DFT calculations were accompanied with experimental results in order to obtain a deeper insight into the electronic structure and vibrational normal modes of complex. Oxidation of sulfides to sulfoxides in one-step was conducted by this complex as catalyst using urea hydrogen peroxide (UHP) in mixture of CH2Cl2/CH3OH (1:1) under air at room temperature. The results show that using this system in oxidation of sulfides, sulfoxides are obtained as the main products, together with variable amounts of sulfones (⩽13%), depending on the nature of the substrate.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide