| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1234465 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2012 | 8 Pages |
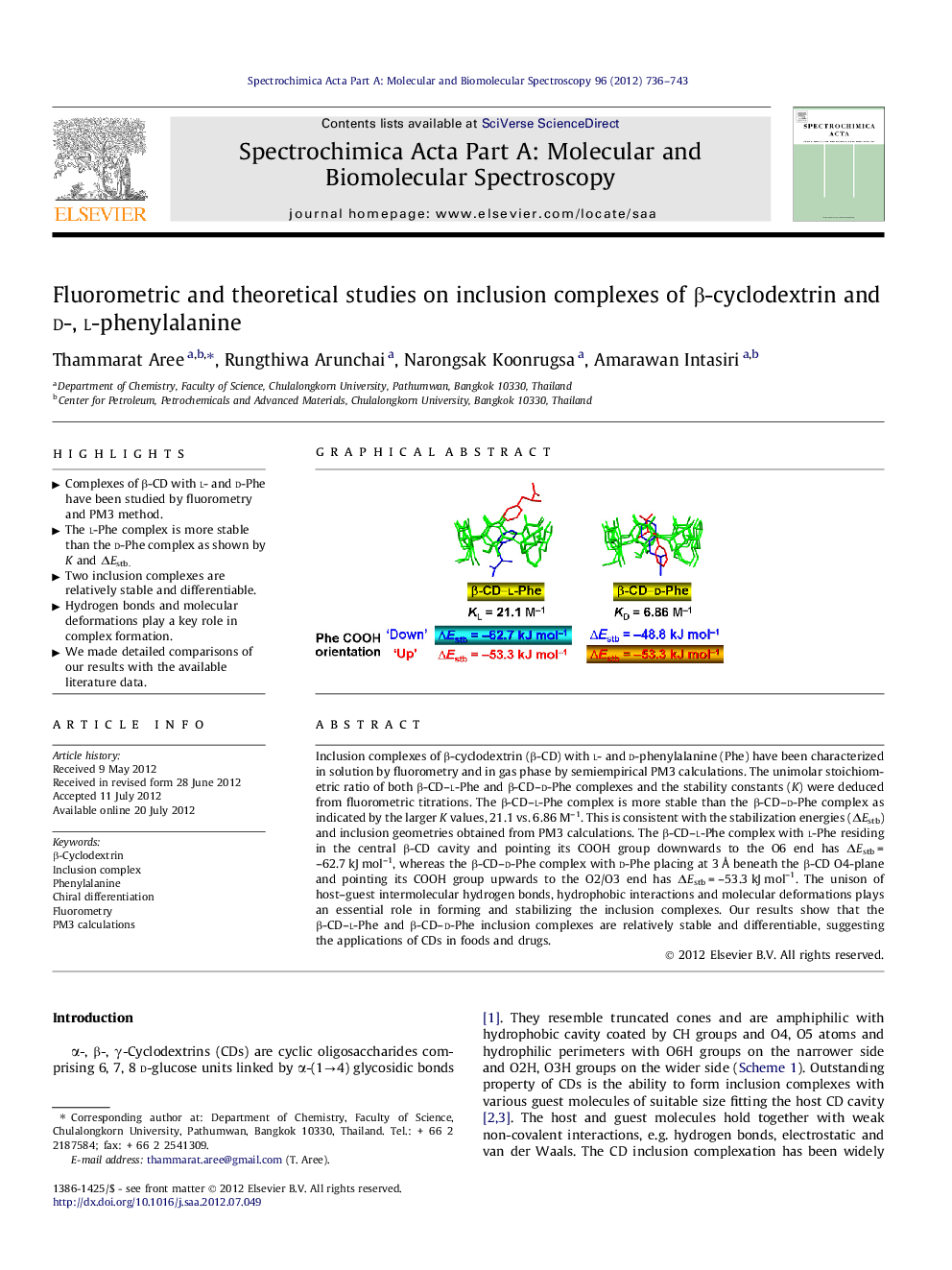
Inclusion complexes of β-cyclodextrin (β-CD) with l- and d-phenylalanine (Phe) have been characterized in solution by fluorometry and in gas phase by semiempirical PM3 calculations. The unimolar stoichiometric ratio of both β-CD–l-Phe and β-CD–d-Phe complexes and the stability constants (K) were deduced from fluorometric titrations. The β-CD–l-Phe complex is more stable than the β-CD–d-Phe complex as indicated by the larger K values, 21.1 vs. 6.86 M–1. This is consistent with the stabilization energies (ΔEstb) and inclusion geometries obtained from PM3 calculations. The β-CD–l-Phe complex with l-Phe residing in the central β-CD cavity and pointing its COOH group downwards to the O6 end has ΔEstb = –62.7 kJ mol–1, whereas the β-CD–d-Phe complex with d-Phe placing at 3 Å beneath the β-CD O4-plane and pointing its COOH group upwards to the O2/O3 end has ΔEstb = –53.3 kJ mol–1. The unison of host–guest intermolecular hydrogen bonds, hydrophobic interactions and molecular deformations plays an essential role in forming and stabilizing the inclusion complexes. Our results show that the β-CD–l-Phe and β-CD–d-Phe inclusion complexes are relatively stable and differentiable, suggesting the applications of CDs in foods and drugs.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slideHighlights► Complexes of β-CD with l- and d-Phe have been studied by fluorometry and PM3 method. ► The l-Phe complex is more stable than the d-Phe complex as shown by K and ΔEstb. ► Two inclusion complexes are relatively stable and differentiable. ► Hydrogen bonds and molecular deformations play a key role in complex formation. ► We made detailed comparisons of our results with the available literature data.