| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1347672 | Tetrahedron: Asymmetry | 2008 | 4 Pages |
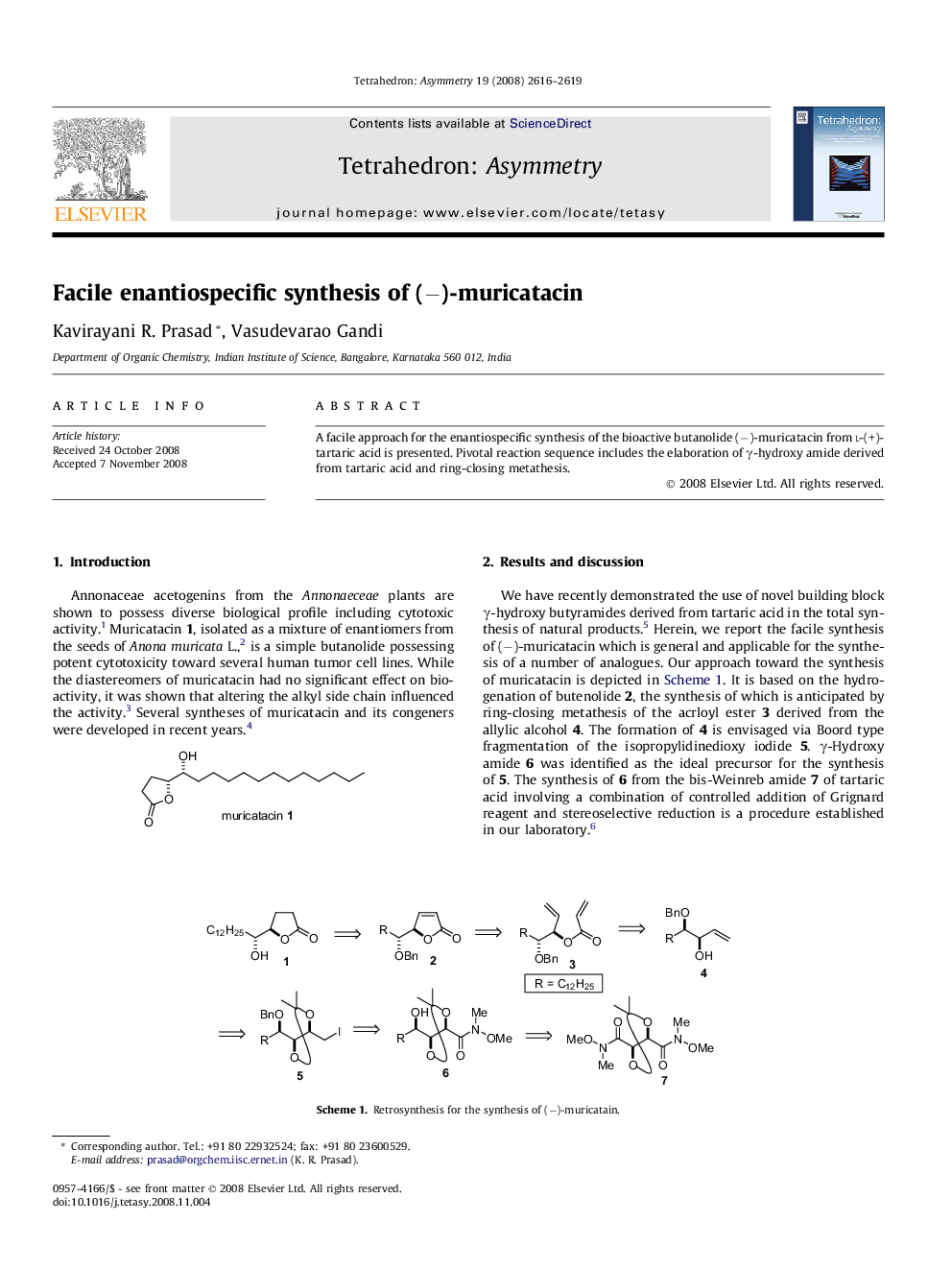
A facile approach for the enantiospecific synthesis of the bioactive butanolide (−)-muricatacin from l-(+)-tartaric acid is presented. Pivotal reaction sequence includes the elaboration of γ-hydroxy amide derived from tartaric acid and ring-closing metathesis.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide
(4R,5R)-N-Methoxy-N,2,2-trimethyl-5-tridecanoyl-1,3-dioxolane-4-carboxamideC21H39NO5[α]D=+4.4[α]D=+4.4 (c 1.1, CHCl3)Source of chirality: l-(+)-tartaric acidAbsolute configuration: (4R,5R)
(4R,5S)-5-((R)-1-Hydroxytridecyl)-N-methoxy-N,2,2-trimethyl-1,3-dioxolane-4-carboxamideC21H41NO5[α]D=-4.3[α]D=-4.3 (c 1.1, CHCl3)Source of chirality: l-(+)-tartaric acidAbsolute configuration: (4R,5S)
(4R,5S)-5-((R)-1-(Benzyloxy)tridecyl)-N-methoxy-N,2,2-trimethyl-1,3-dioxolane-4-carboxamideC28H47NO5[α]D=+6.5[α]D=+6.5 (c 1.4, CHCl3)Source of chirality: l-(+)-tartaric acidAbsolute configuration: (4R,5S)
(3R,4R)-4-(Benzyloxy)hexadec-1-en-3-olC23H38O2[α]D=-3.6[α]D=-3.6 (c 1.7, CHCl3)Source of chirality: l-(+)-tartaric acidAbsolute configuration: (3R,4R)
(R)-5-((R)-1-(Benzyloxy)tridecyl)furan-2(5H)-oneC24H36O3[α]D=+104.5[α]D=+104.5 (c 1.1, CHCl3)Source of chirality: l-(+)-tartaric acidAbsolute configuration: (4R,5R)
(−)-MuricatacinC17H32O3[α]D=-23.9[α]D=-23.9 (c 0.8, CHCl3)Source of chirality: l-(+)-tartaric acidAbsolute configuration: (4R,5R)