| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1387979 | Carbohydrate Research | 2010 | 5 Pages |
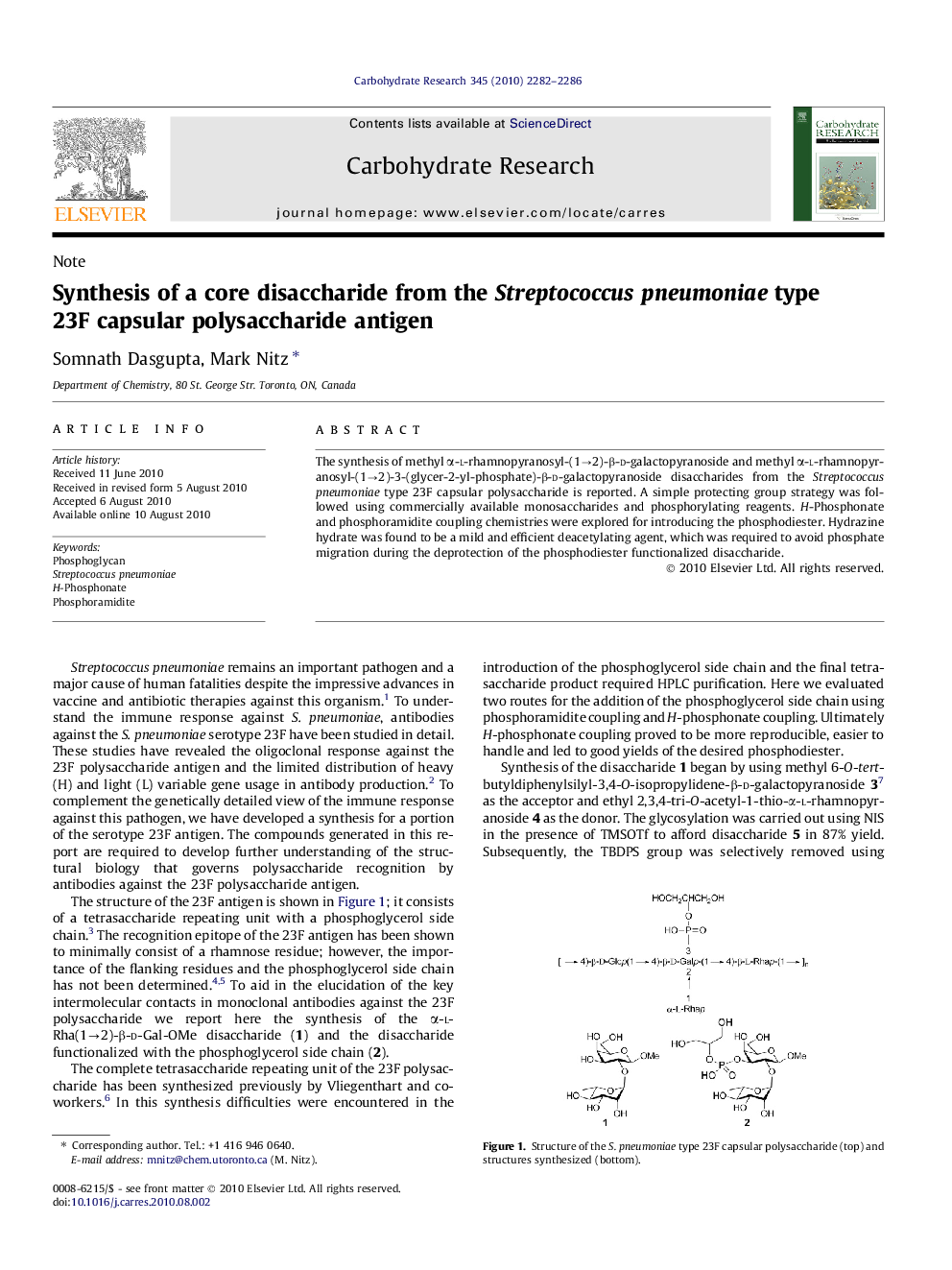
The synthesis of methyl α-l-rhamnopyranosyl-(1→2)-β-d-galactopyranoside and methyl α-l-rhamnopyranosyl-(1→2)-3-(glycer-2-yl-phosphate)-β-d-galactopyranoside disaccharides from the Streptococcuspneumoniae type 23F capsular polysaccharide is reported. A simple protecting group strategy was followed using commercially available monosaccharides and phosphorylating reagents. H-Phosphonate and phosphoramidite coupling chemistries were explored for introducing the phosphodiester. Hydrazine hydrate was found to be a mild and efficient deacetylating agent, which was required to avoid phosphate migration during the deprotection of the phosphodiester functionalized disaccharide.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide