| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1564712 | Journal of Nuclear Materials | 2016 | 7 Pages |
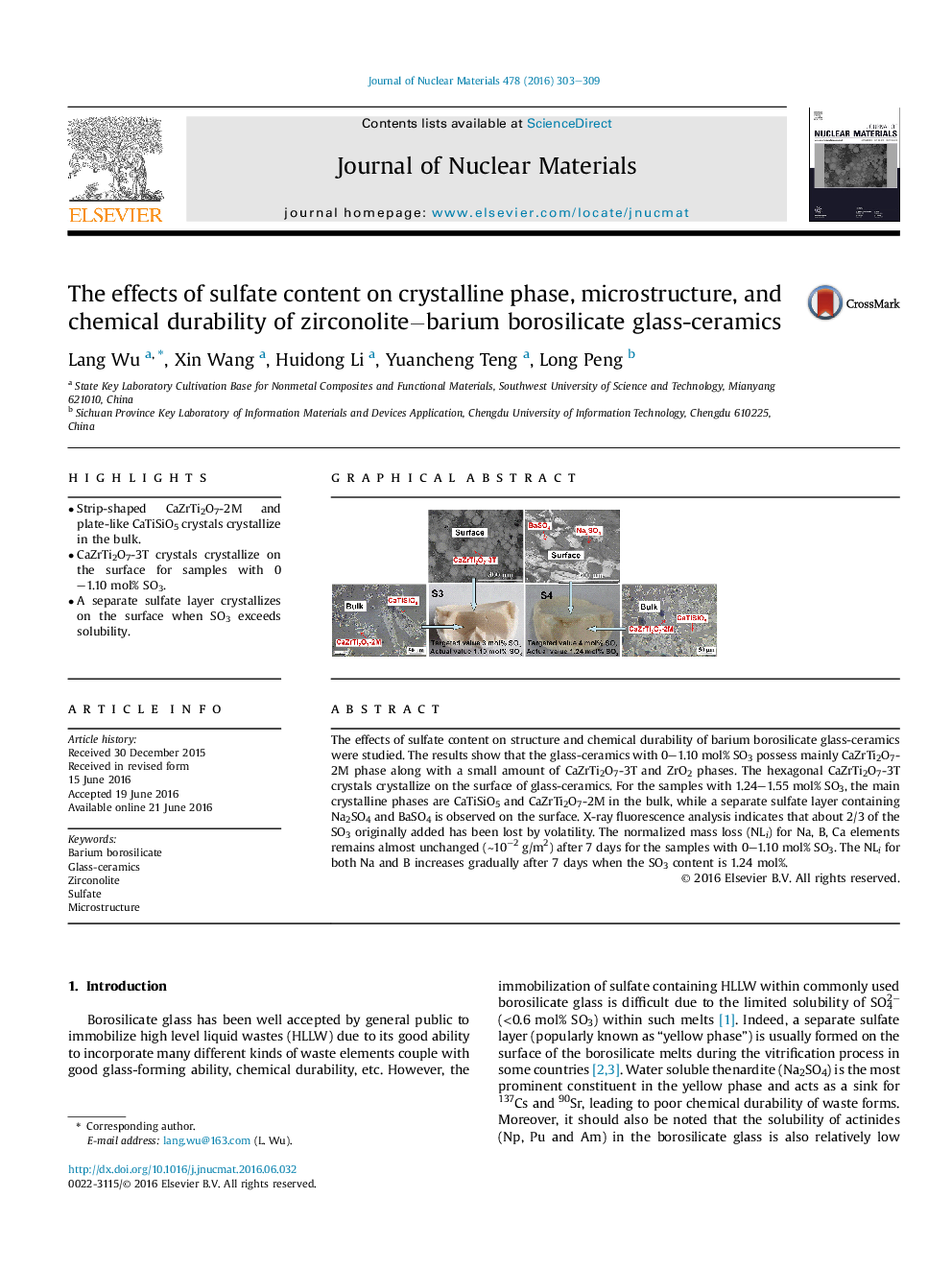
•Strip-shaped CaZrTi2O7-2M and plate-like CaTiSiO5 crystals crystallize in the bulk.•CaZrTi2O7-3T crystals crystallize on the surface for samples with 0–1.10 mol% SO3.•A separate sulfate layer crystallizes on the surface when SO3 exceeds solubility.
The effects of sulfate content on structure and chemical durability of barium borosilicate glass-ceramics were studied. The results show that the glass-ceramics with 0–1.10 mol% SO3 possess mainly CaZrTi2O7-2M phase along with a small amount of CaZrTi2O7-3T and ZrO2 phases. The hexagonal CaZrTi2O7-3T crystals crystallize on the surface of glass-ceramics. For the samples with 1.24–1.55 mol% SO3, the main crystalline phases are CaTiSiO5 and CaZrTi2O7-2M in the bulk, while a separate sulfate layer containing Na2SO4 and BaSO4 is observed on the surface. X-ray fluorescence analysis indicates that about 2/3 of the SO3 originally added has been lost by volatility. The normalized mass loss (NLi) for Na, B, Ca elements remains almost unchanged (∼10−2 g/m2) after 7 days for the samples with 0–1.10 mol% SO3. The NLi for both Na and B increases gradually after 7 days when the SO3 content is 1.24 mol%.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide