| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5220388 | Tetrahedron | 2012 | 4 Pages |
Abstract
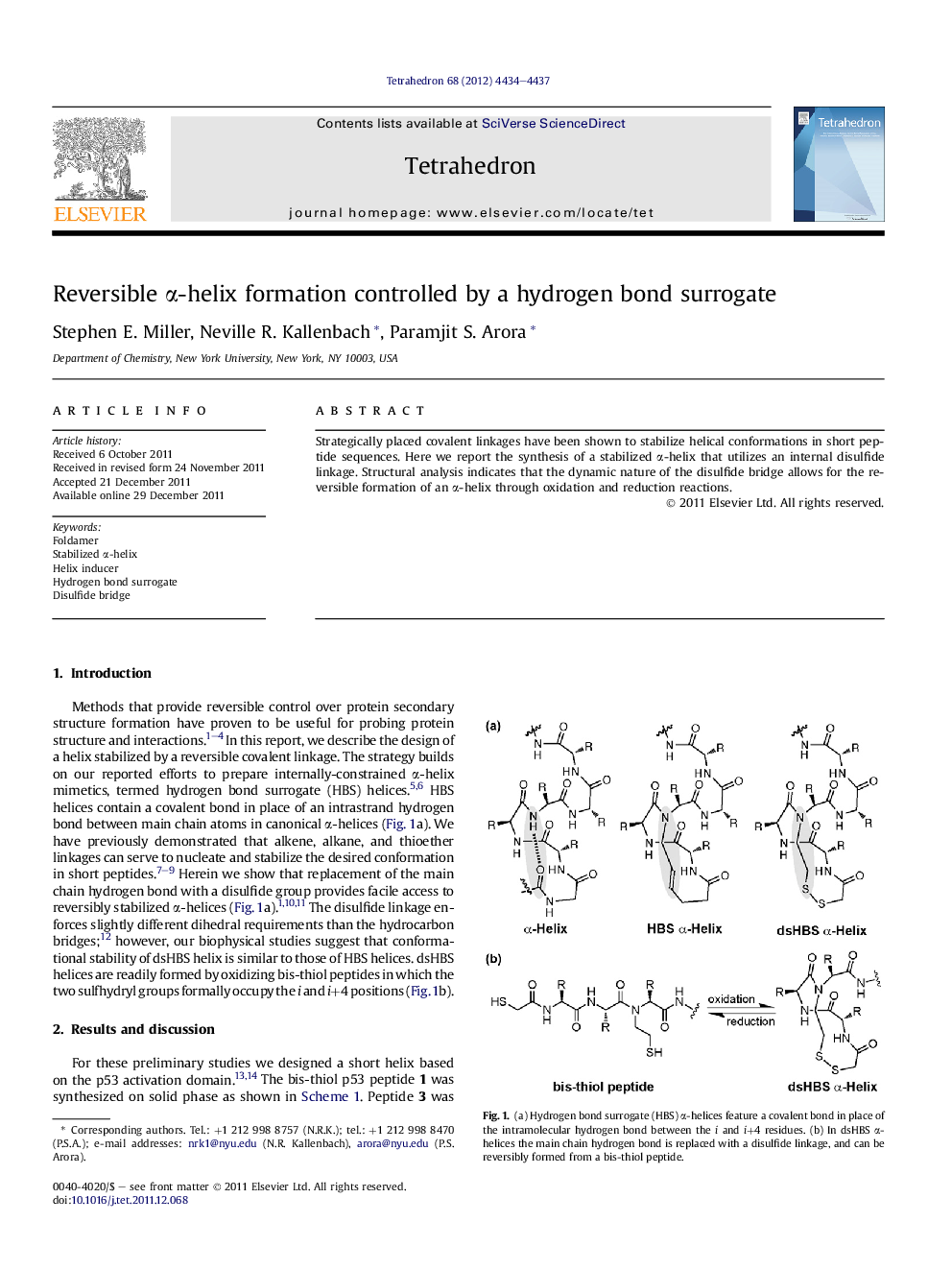
Strategically placed covalent linkages have been shown to stabilize helical conformations in short peptide sequences. Here we report the synthesis of a stabilized α-helix that utilizes an internal disulfide linkage. Structural analysis indicates that the dynamic nature of the disulfide bridge allows for the reversible formation of an α-helix through oxidation and reduction reactions.
Graphical abstractDownload full-size image
Keywords
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Stephen E. Miller, Neville R. Kallenbach, Paramjit S. Arora,