| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5220986 | Tetrahedron | 2011 | 5 Pages |
Abstract
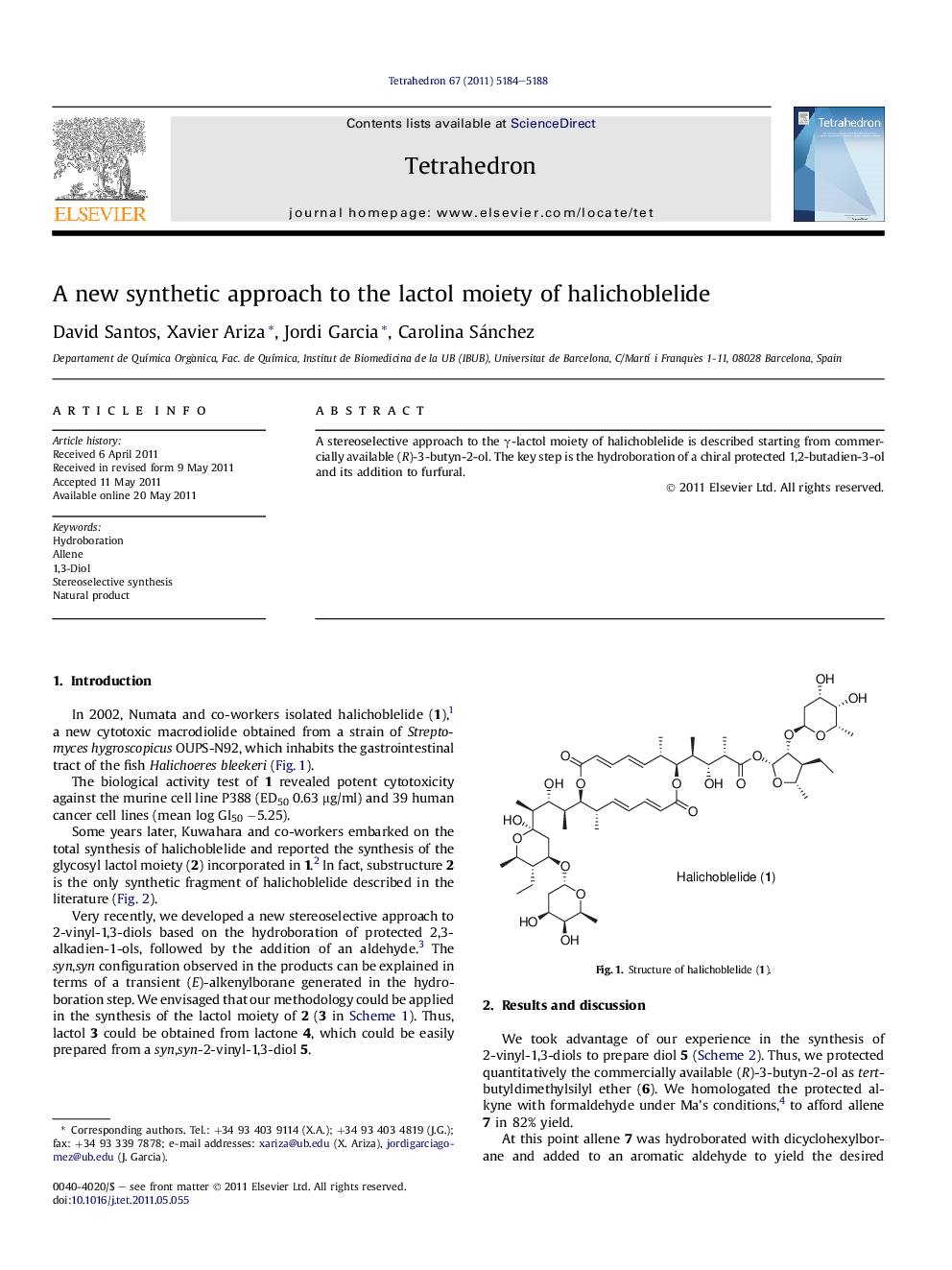
A stereoselective approach to the γ-lactol moiety of halichoblelide is described starting from commercially available (R)-3-butyn-2-ol. The key step is the hydroboration of a chiral protected 1,2-butadien-3-ol and its addition to furfural.
Graphical abstractDownload full-size image
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
David Santos, Xavier Ariza, Jordi Garcia, Carolina Sánchez,