| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5222816 | Tetrahedron | 2009 | 9 Pages |
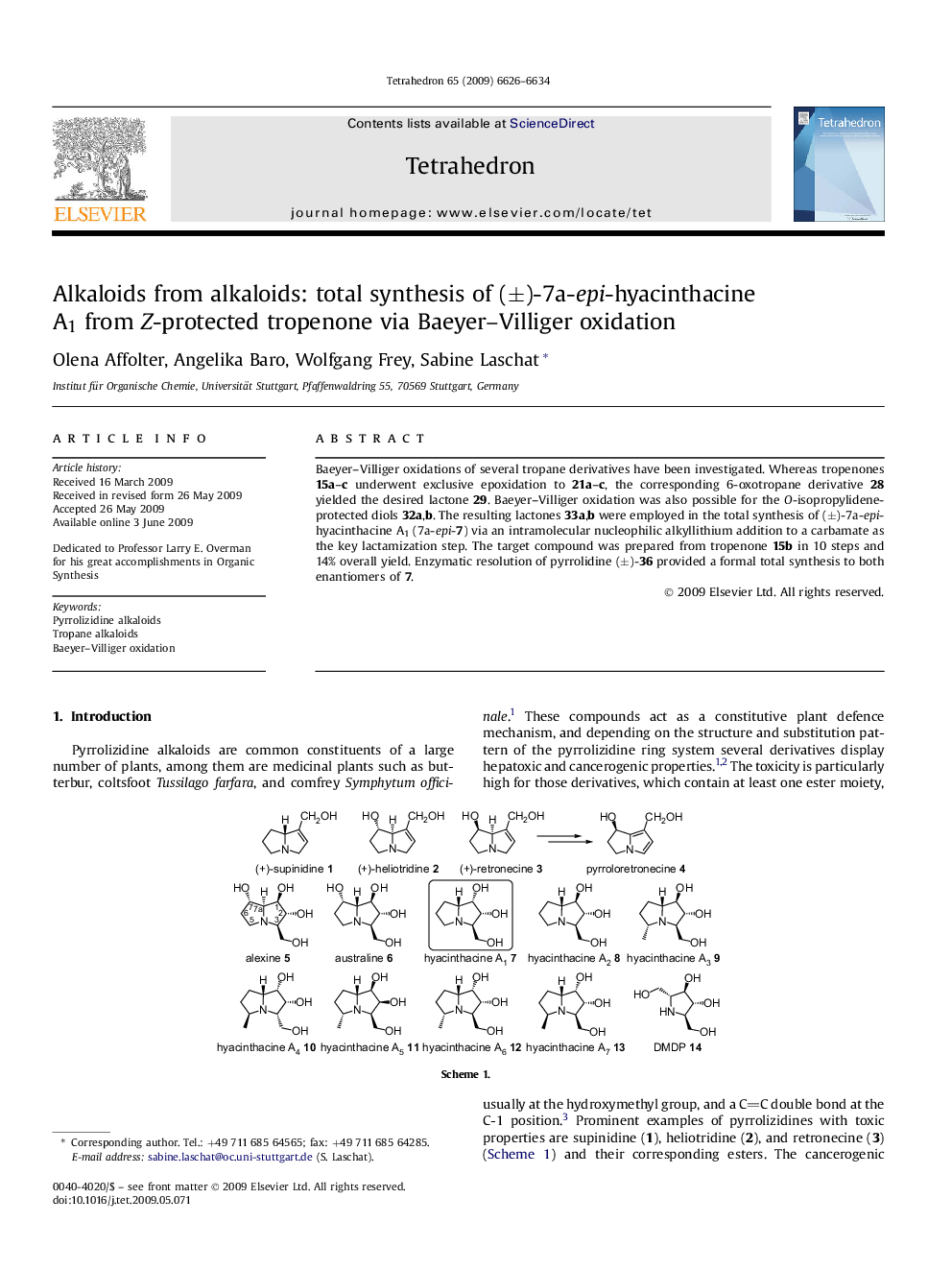
Baeyer–Villiger oxidations of several tropane derivatives have been investigated. Whereas tropenones 15a–c underwent exclusive epoxidation to 21a–c, the corresponding 6-oxotropane derivative 28 yielded the desired lactone 29. Baeyer–Villiger oxidation was also possible for the O-isopropylidene-protected diols 32a,b. The resulting lactones 33a,b were employed in the total synthesis of (±)-7a-epi-hyacinthacine A1 (7a-epi-7) via an intramolecular nucleophilic alkyllithium addition to a carbamate as the key lactamization step. The target compound was prepared from tropenone 15b in 10 steps and 14% overall yield. Enzymatic resolution of pyrrolidine (±)-36 provided a formal total synthesis to both enantiomers of 7.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide