| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5271693 | Tetrahedron Letters | 2013 | 4 Pages |
Abstract
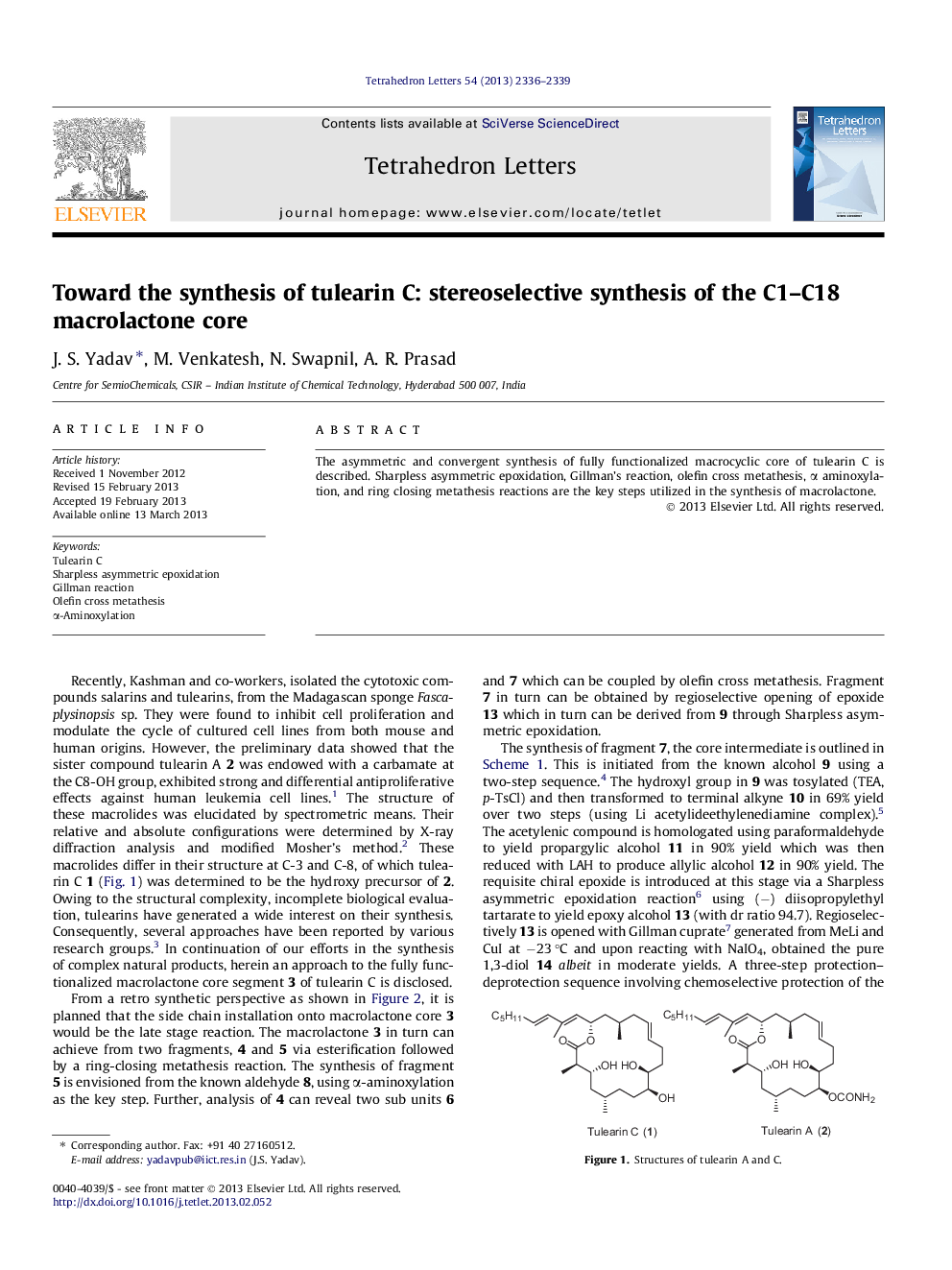
The asymmetric and convergent synthesis of fully functionalized macrocyclic core of tulearin C is described. Sharpless asymmetric epoxidation, Gillman's reaction, olefin cross metathesis, α aminoxylation, and ring closing metathesis reactions are the key steps utilized in the synthesis of macrolactone.
Graphical abstractDownload full-size image
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
J.S. Yadav, M. Venkatesh, N. Swapnil, A.R. Prasad,