| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5280044 | Tetrahedron Letters | 2005 | 5 Pages |
Abstract
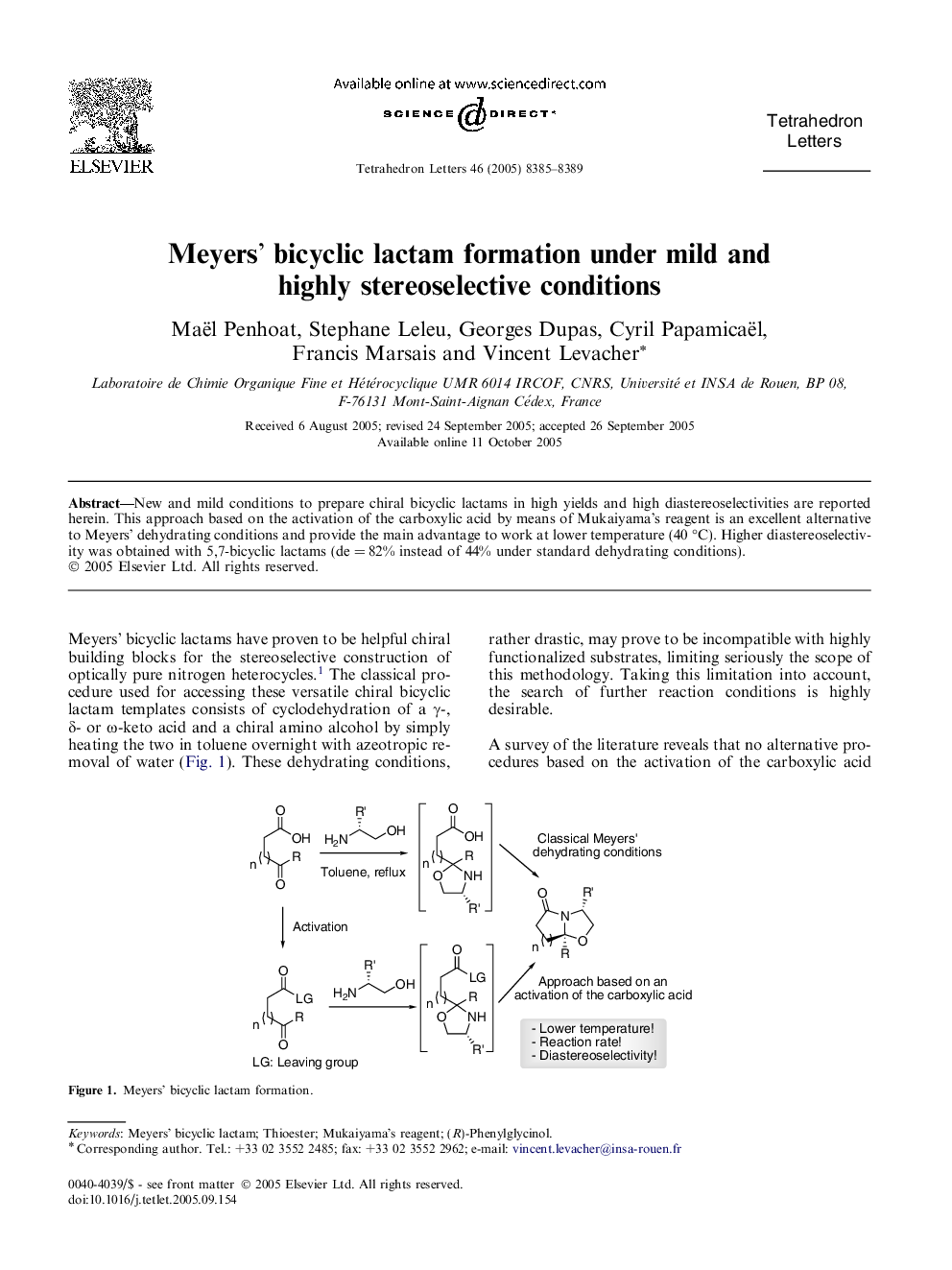
New and mild conditions to prepare chiral bicyclic lactams in high yields and high diastereoselectivities are reported herein. This approach based on the activation of the carboxylic acid by means of Mukaiyama's reagent is an excellent alternative to Meyers' dehydrating conditions and provide the main advantage to work at lower temperature (40 °C). Higher diastereoselectivity was obtained with 5,7-bicyclic lactams (de = 82% instead of 44% under standard dehydrating conditions).
Graphical abstractDownload full-size image
Keywords
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Maël Penhoat, Stephane Leleu, Georges Dupas, Cyril Papamicaël, Francis Marsais, Vincent Levacher,