| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5282153 | Tetrahedron Letters | 2007 | 4 Pages |
Abstract
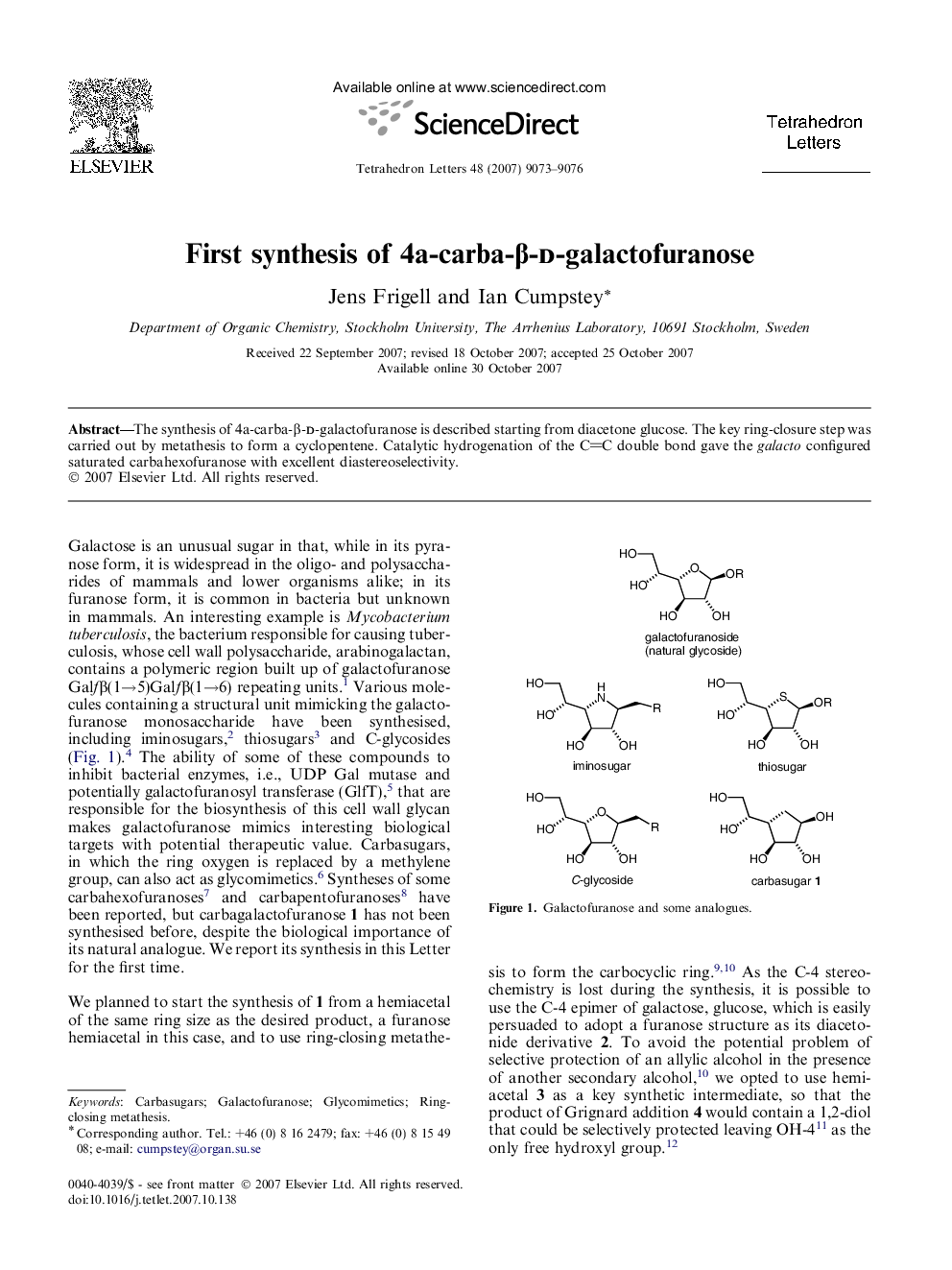
The synthesis of 4a-carba-β-d-galactofuranose is described starting from diacetone glucose. The key ring-closure step was carried out by metathesis to form a cyclopentene. Catalytic hydrogenation of the CC double bond gave the galacto configured saturated carbahexofuranose with excellent diastereoselectivity.
Graphical abstractDownload full-size image
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Jens Frigell, Ian Cumpstey,