| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 53123 | Catalysis Communications | 2007 | 4 Pages |
Abstract
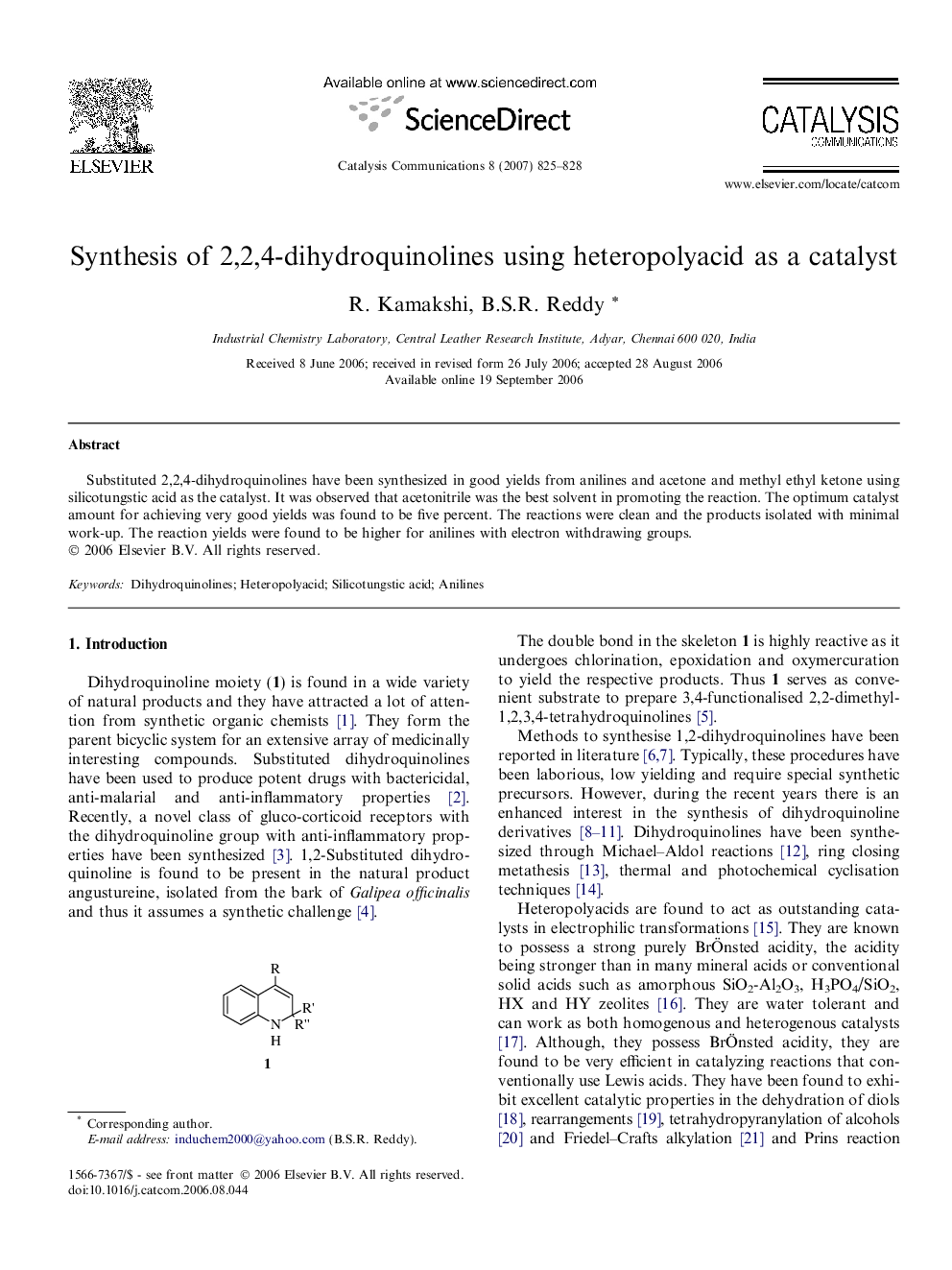
Substituted 2,2,4-dihydroquinolines have been synthesized in good yields from anilines and acetone and methyl ethyl ketone using silicotungstic acid as the catalyst. It was observed that acetonitrile was the best solvent in promoting the reaction. The optimum catalyst amount for achieving very good yields was found to be five percent. The reactions were clean and the products isolated with minimal work-up. The reaction yields were found to be higher for anilines with electron withdrawing groups.
Related Topics
Physical Sciences and Engineering
Chemical Engineering
Catalysis
Authors
R. Kamakshi, B.S.R. Reddy,