| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5371240 | Biophysical Chemistry | 2012 | 6 Pages |
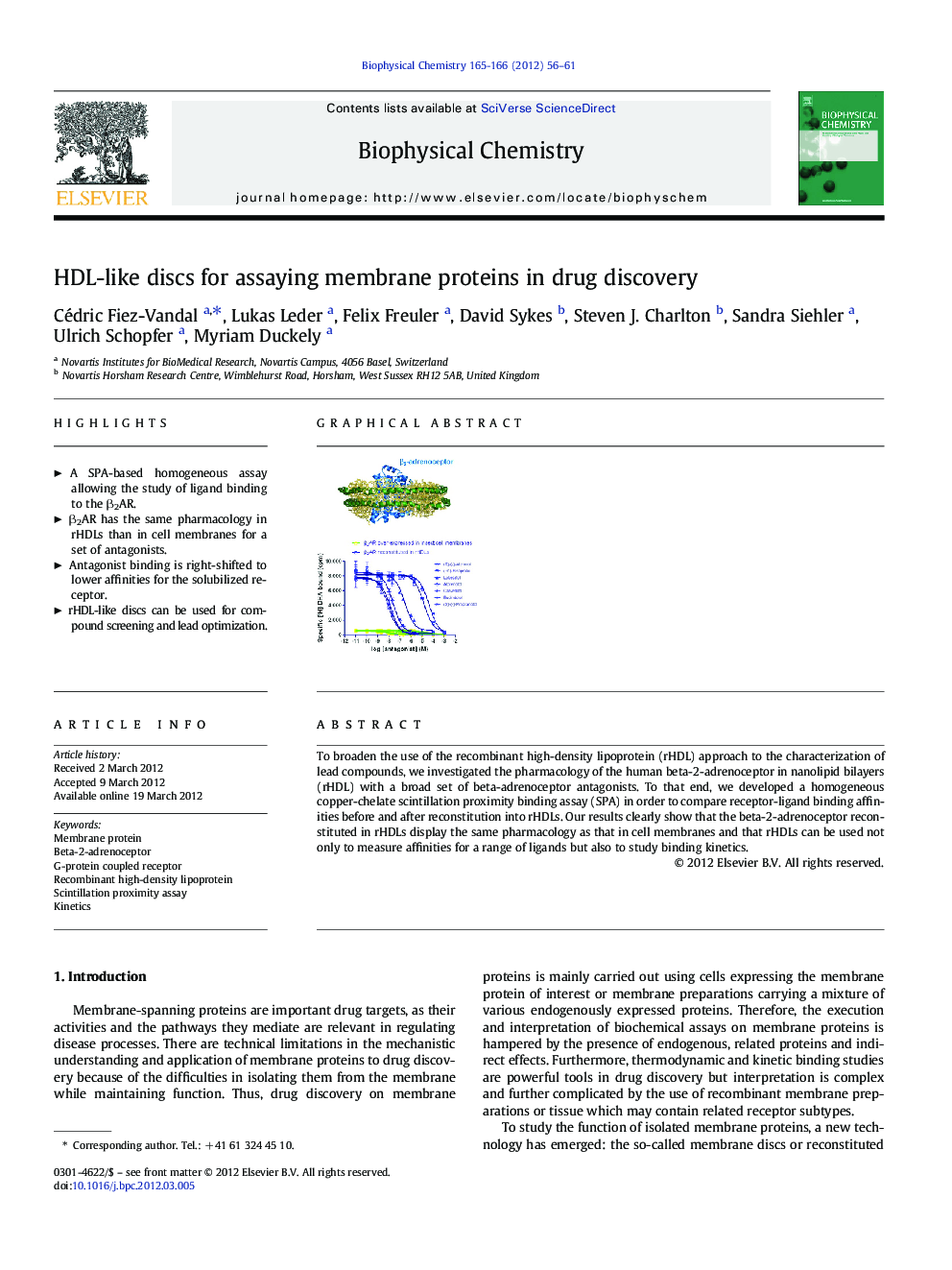
To broaden the use of the recombinant high-density lipoprotein (rHDL) approach to the characterization of lead compounds, we investigated the pharmacology of the human beta-2-adrenoceptor in nanolipid bilayers (rHDL) with a broad set of beta-adrenoceptor antagonists. To that end, we developed a homogeneous copper-chelate scintillation proximity binding assay (SPA) in order to compare receptor-ligand binding affinities before and after reconstitution into rHDLs. Our results clearly show that the beta-2-adrenoceptor reconstituted in rHDLs display the same pharmacology as that in cell membranes and that rHDLs can be used not only to measure affinities for a range of ligands but also to study binding kinetics.
Graphical abstractDownload full-size imageHighlights⺠A SPA-based homogeneous assay allowing the study of ligand binding to the β2AR. ⺠β2AR has the same pharmacology in rHDLs than in cell membranes for a set of antagonists. ⺠Antagonist binding is right-shifted to lower affinities for the solubilized receptor. ⺠rHDL-like discs can be used for compound screening and lead optimization.