| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 7672841 | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy | 2013 | 7 Pages |
Abstract
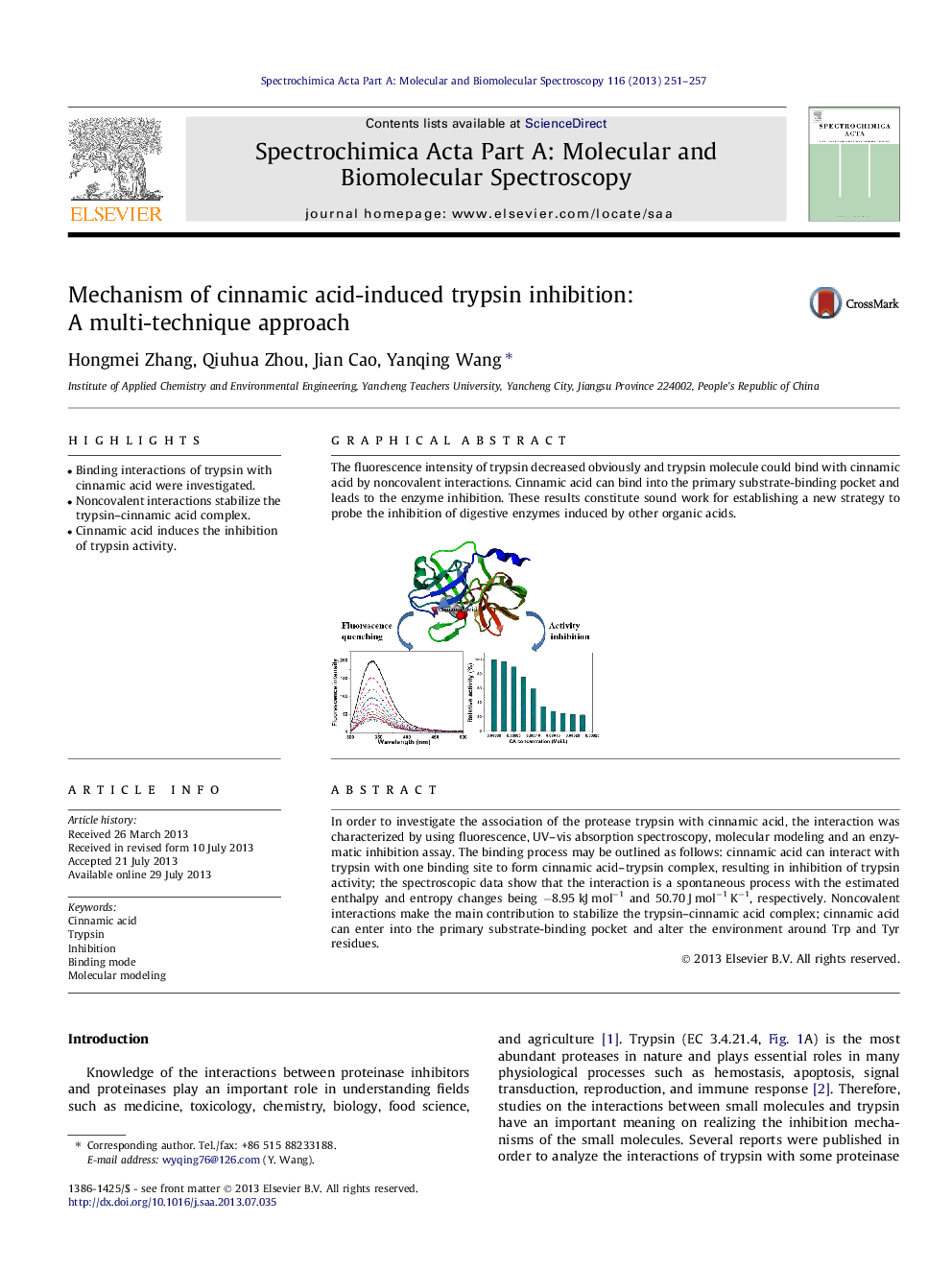
The fluorescence intensity of trypsin decreased obviously and trypsin molecule could bind with cinnamic acid by noncovalent interactions. Cinnamic acid can bind into the primary substrate-binding pocket and leads to the enzyme inhibition. These results constitute sound work for establishing a new strategy to probe the inhibition of digestive enzymes induced by other organic acids.
Related Topics
Physical Sciences and Engineering
Chemistry
Analytical Chemistry
Authors
Hongmei Zhang, Qiuhua Zhou, Jian Cao, Yanqing Wang,