| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1402791 | Journal of Molecular Structure | 2014 | 9 Pages |
•Comprehensive PED assignment for 1-methyl-4-nitropyrazole is reported.•IR, Raman, 1H NMR, 1H–1H NMR and 13C NMR and cyclic voltamperometry methods are used.•The fully anharmonic IR and Raman spectra are presented for the first time.•Detailed NBO analysis has been done in order to explain the stabilizing interaction.•The stability arising from hyper conjugative interaction is demonstrated.
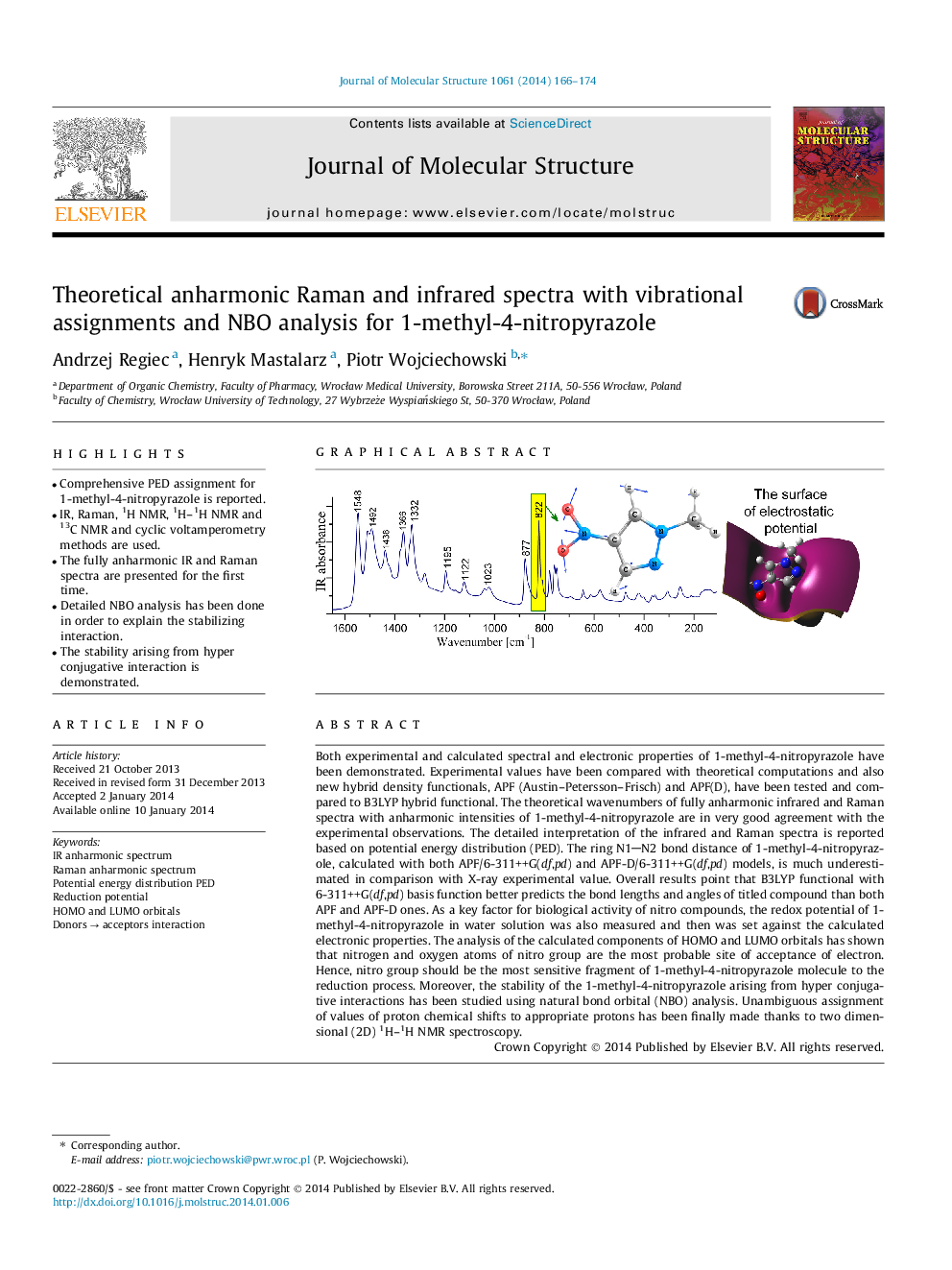
Both experimental and calculated spectral and electronic properties of 1-methyl-4-nitropyrazole have been demonstrated. Experimental values have been compared with theoretical computations and also new hybrid density functionals, APF (Austin–Petersson–Frisch) and APF(D), have been tested and compared to B3LYP hybrid functional. The theoretical wavenumbers of fully anharmonic infrared and Raman spectra with anharmonic intensities of 1-methyl-4-nitropyrazole are in very good agreement with the experimental observations. The detailed interpretation of the infrared and Raman spectra is reported based on potential energy distribution (PED). The ring N1N2 bond distance of 1-methyl-4-nitropyrazole, calculated with both APF/6-311++G(df,pd) and APF-D/6-311++G(df,pd) models, is much underestimated in comparison with X-ray experimental value. Overall results point that B3LYP functional with 6-311++G(df,pd) basis function better predicts the bond lengths and angles of titled compound than both APF and APF-D ones. As a key factor for biological activity of nitro compounds, the redox potential of 1-methyl-4-nitropyrazole in water solution was also measured and then was set against the calculated electronic properties. The analysis of the calculated components of HOMO and LUMO orbitals has shown that nitrogen and oxygen atoms of nitro group are the most probable site of acceptance of electron. Hence, nitro group should be the most sensitive fragment of 1-methyl-4-nitropyrazole molecule to the reduction process. Moreover, the stability of the 1-methyl-4-nitropyrazole arising from hyper conjugative interactions has been studied using natural bond orbital (NBO) analysis. Unambiguous assignment of values of proton chemical shifts to appropriate protons has been finally made thanks to two dimensional (2D) 1H–1H NMR spectroscopy.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide