| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1404898 | Journal of Molecular Structure | 2015 | 5 Pages |
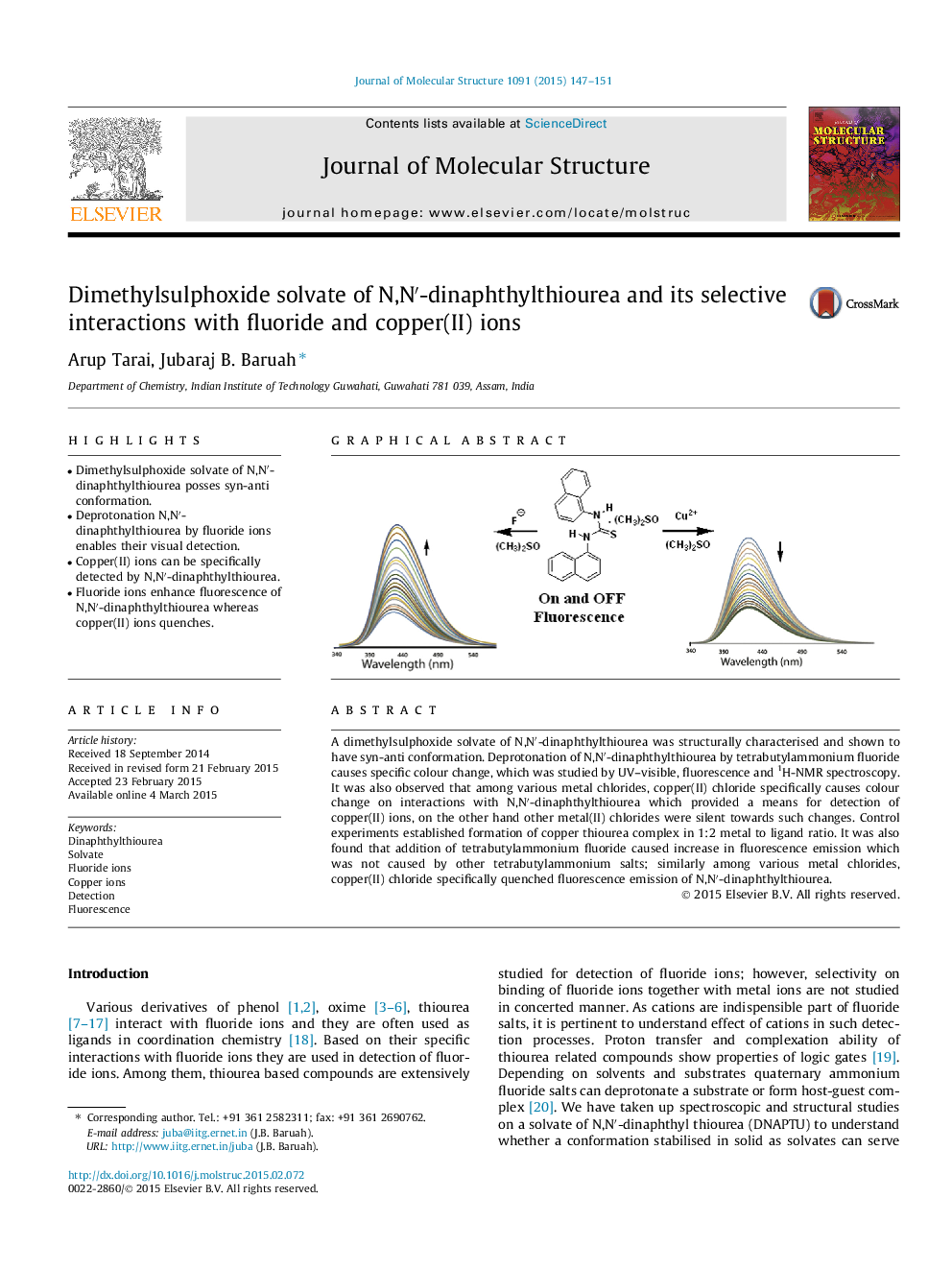
•Dimethylsulphoxide solvate of N,N′-dinaphthylthiourea posses syn-anti conformation.•Deprotonation N,N′-dinaphthylthiourea by fluoride ions enables their visual detection.•Copper(II) ions can be specifically detected by N,N′-dinaphthylthiourea.•Fluoride ions enhance fluorescence of N,N′-dinaphthylthiourea whereas copper(II) ions quenches.
A dimethylsulphoxide solvate of N,N′-dinaphthylthiourea was structurally characterised and shown to have syn-anti conformation. Deprotonation of N,N′-dinaphthylthiourea by tetrabutylammonium fluoride causes specific colour change, which was studied by UV–visible, fluorescence and 1H-NMR spectroscopy. It was also observed that among various metal chlorides, copper(II) chloride specifically causes colour change on interactions with N,N′-dinaphthylthiourea which provided a means for detection of copper(II) ions, on the other hand other metal(II) chlorides were silent towards such changes. Control experiments established formation of copper thiourea complex in 1:2 metal to ligand ratio. It was also found that addition of tetrabutylammonium fluoride caused increase in fluorescence emission which was not caused by other tetrabutylammonium salts; similarly among various metal chlorides, copper(II) chloride specifically quenched fluorescence emission of N,N′-dinaphthylthiourea.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide