| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 5130593 | Analytica Chimica Acta | 2017 | 6 Pages |
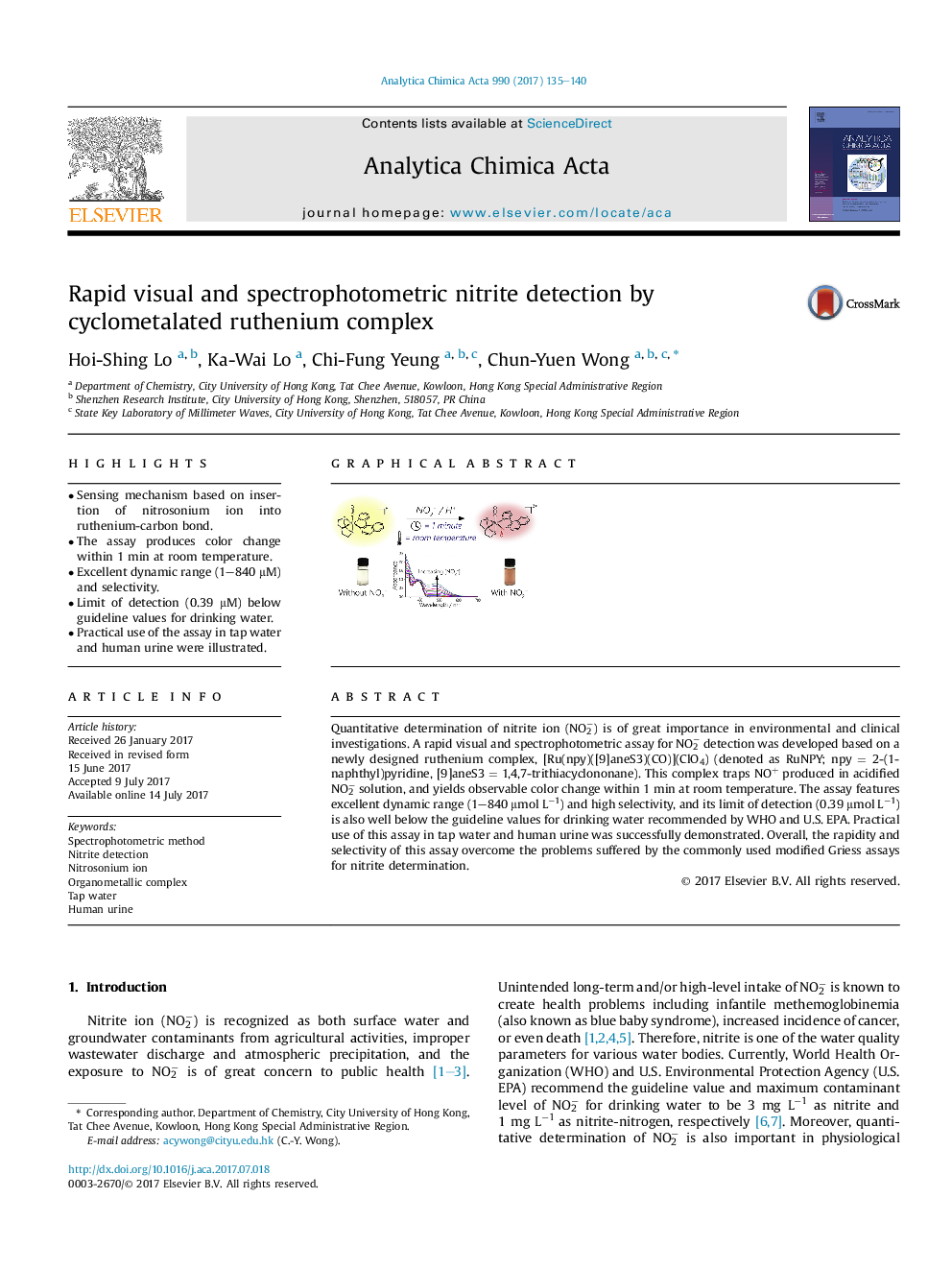
•Sensing mechanism based on insertion of nitrosonium ion into ruthenium-carbon bond.•The assay produces color change within 1 min at room temperature.•Excellent dynamic range (1–840 μM) and selectivity.•Limit of detection (0.39 μM) below guideline values for drinking water.•Practical use of the assay in tap water and human urine were illustrated.
Quantitative determination of nitrite ion (NO2−) is of great importance in environmental and clinical investigations. A rapid visual and spectrophotometric assay for NO2− detection was developed based on a newly designed ruthenium complex, [Ru(npy)([9]aneS3)(CO)](ClO4) (denoted as RuNPY; npy = 2-(1-naphthyl)pyridine, [9]aneS3 = 1,4,7-trithiacyclononane). This complex traps NO+ produced in acidified NO2− solution, and yields observable color change within 1 min at room temperature. The assay features excellent dynamic range (1–840 μmol L−1) and high selectivity, and its limit of detection (0.39 μmol L−1) is also well below the guideline values for drinking water recommended by WHO and U.S. EPA. Practical use of this assay in tap water and human urine was successfully demonstrated. Overall, the rapidity and selectivity of this assay overcome the problems suffered by the commonly used modified Griess assays for nitrite determination.
Graphical abstractFigure optionsDownload full-size imageDownload high-quality image (206 K)Download as PowerPoint slide