| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 10594091 | Bioorganic & Medicinal Chemistry Letters | 2011 | 5 Pages |
Abstract
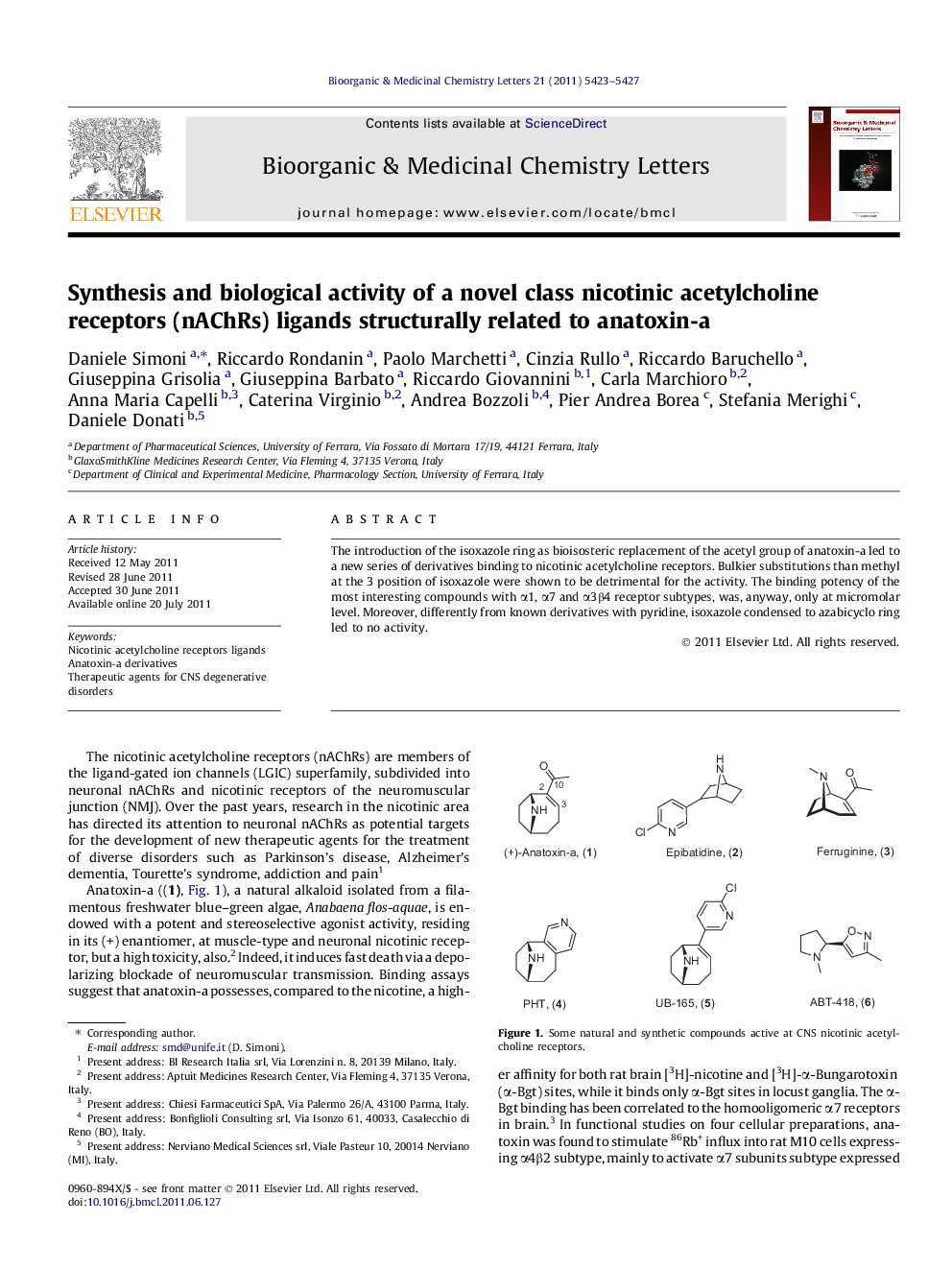
The introduction of the isoxazole ring as bioisosteric replacement of the acetyl group of anatoxin-a led to a new series of derivatives binding to nicotinic acetylcholine receptors. Bulkier substitutions than methyl at the 3 position of isoxazole were shown to be detrimental for the activity. The binding potency of the most interesting compounds with α1, α7 and α3β4 receptor subtypes, was, anyway, only at micromolar level. Moreover, differently from known derivatives with pyridine, isoxazole condensed to azabicyclo ring led to no activity.
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Daniele Simoni, Riccardo Rondanin, Paolo Marchetti, Cinzia Rullo, Riccardo Baruchello, Giuseppina Grisolia, Giuseppina Barbato, Riccardo Giovannini, Carla Marchioro, Anna Maria Capelli, Caterina Virginio, Andrea Bozzoli, Pier Andrea Borea,