| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1284364 | Journal of Power Sources | 2013 | 8 Pages |
•Ce–La solid solution supported Rh catalyst shows high activity for H2 production.•The catalyst also shows low CO selectivity and good stability.•The performance of the catalysts varied depending on urea/metal ratio during preparation.
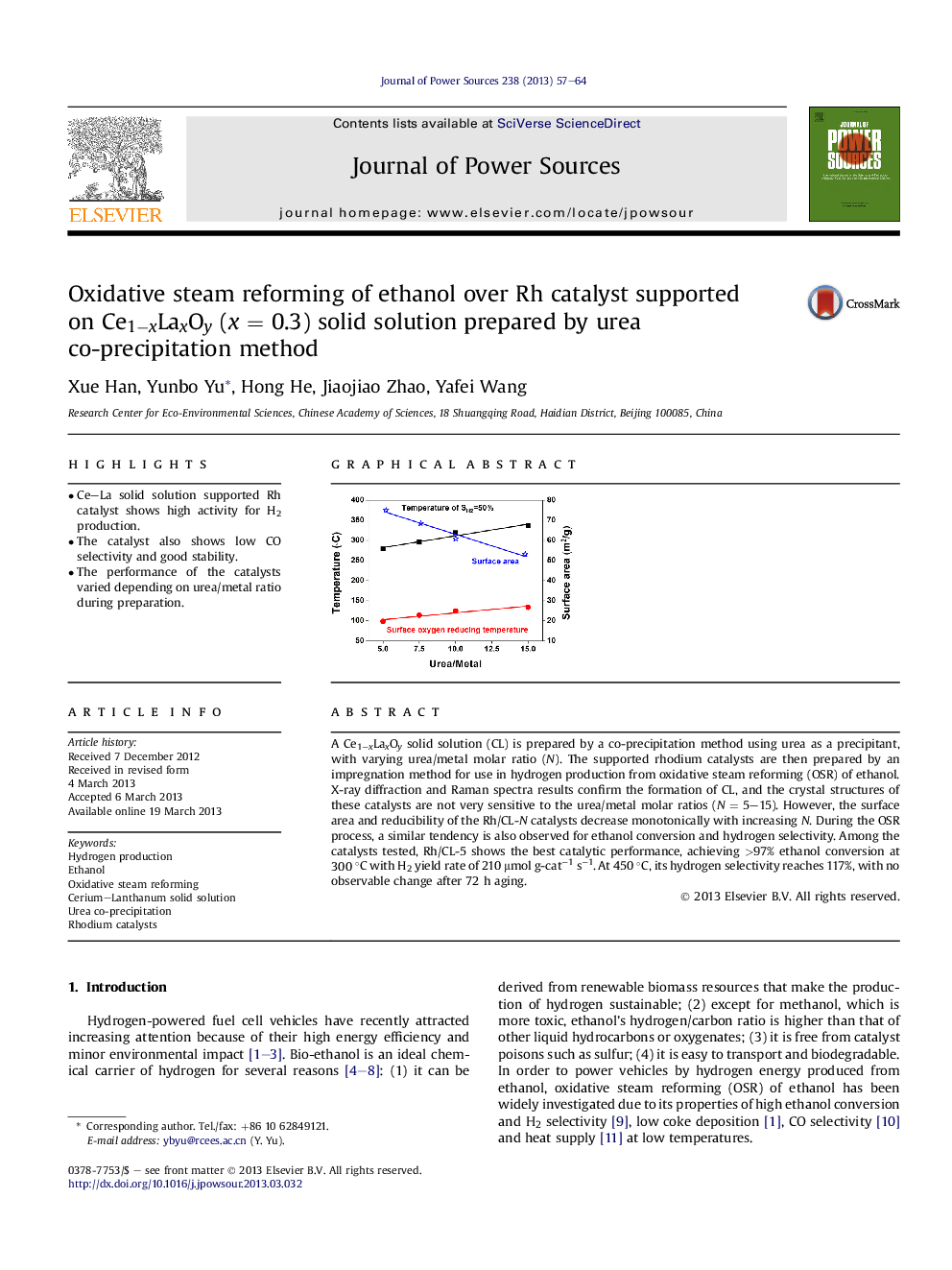
A Ce1−xLaxOy solid solution (CL) is prepared by a co-precipitation method using urea as a precipitant, with varying urea/metal molar ratio (N). The supported rhodium catalysts are then prepared by an impregnation method for use in hydrogen production from oxidative steam reforming (OSR) of ethanol. X-ray diffraction and Raman spectra results confirm the formation of CL, and the crystal structures of these catalysts are not very sensitive to the urea/metal molar ratios (N = 5–15). However, the surface area and reducibility of the Rh/CL-N catalysts decrease monotonically with increasing N. During the OSR process, a similar tendency is also observed for ethanol conversion and hydrogen selectivity. Among the catalysts tested, Rh/CL-5 shows the best catalytic performance, achieving >97% ethanol conversion at 300 °C with H2 yield rate of 210 μmol g-cat−1 s−1. At 450 °C, its hydrogen selectivity reaches 117%, with no observable change after 72 h aging.
Graphical abstractCe1−xLaxOy solid solution-supported Rh catalysts exhibit high activity and long term for oxidative steam reforming of ethanol to H2 at low temperatures. The catalytic performance is greatly affect by the urea/metal molar ratio during the preparation of supports by urea co-precipitation.Figure optionsDownload full-size imageDownload as PowerPoint slide