| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1372487 | Bioorganic & Medicinal Chemistry Letters | 2010 | 4 Pages |
Abstract
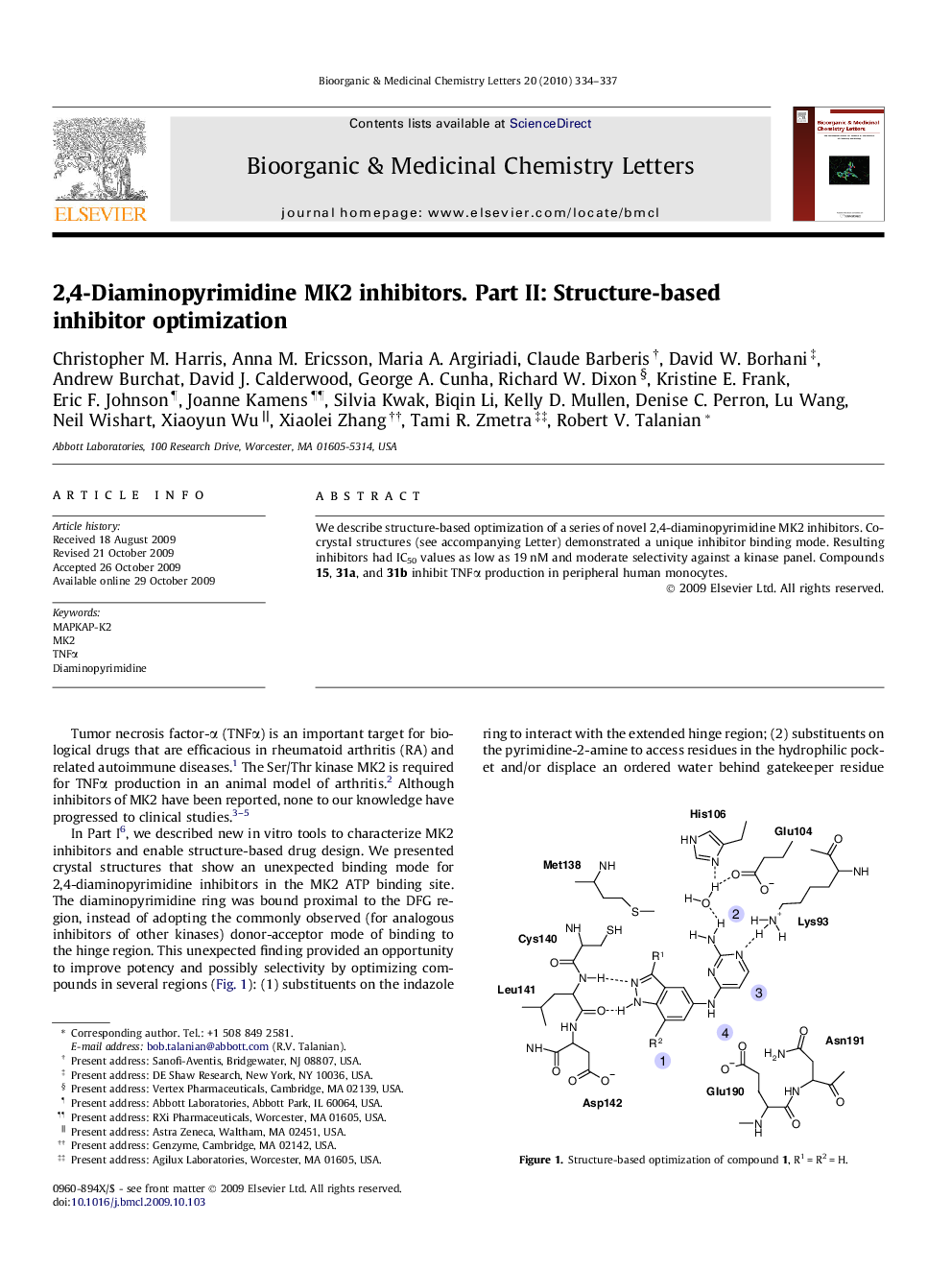
We describe structure-based optimization of a series of novel 2,4-diaminopyrimidine MK2 inhibitors. Co-crystal structures (see accompanying Letter) demonstrated a unique inhibitor binding mode. Resulting inhibitors had IC50 values as low as 19 nM and moderate selectivity against a kinase panel. Compounds 15, 31a, and 31b inhibit TNFα production in peripheral human monocytes.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide
Keywords
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Christopher M. Harris, Anna M. Ericsson, Maria A. Argiriadi, Claude Barberis, David W. Borhani, Andrew Burchat, David J. Calderwood, George A. Cunha, Richard W. Dixon, Kristine E. Frank, Eric F. Johnson, Joanne Kamens, Silvia Kwak, Biqin Li,