| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1374622 | Bioorganic & Medicinal Chemistry Letters | 2010 | 4 Pages |
Abstract
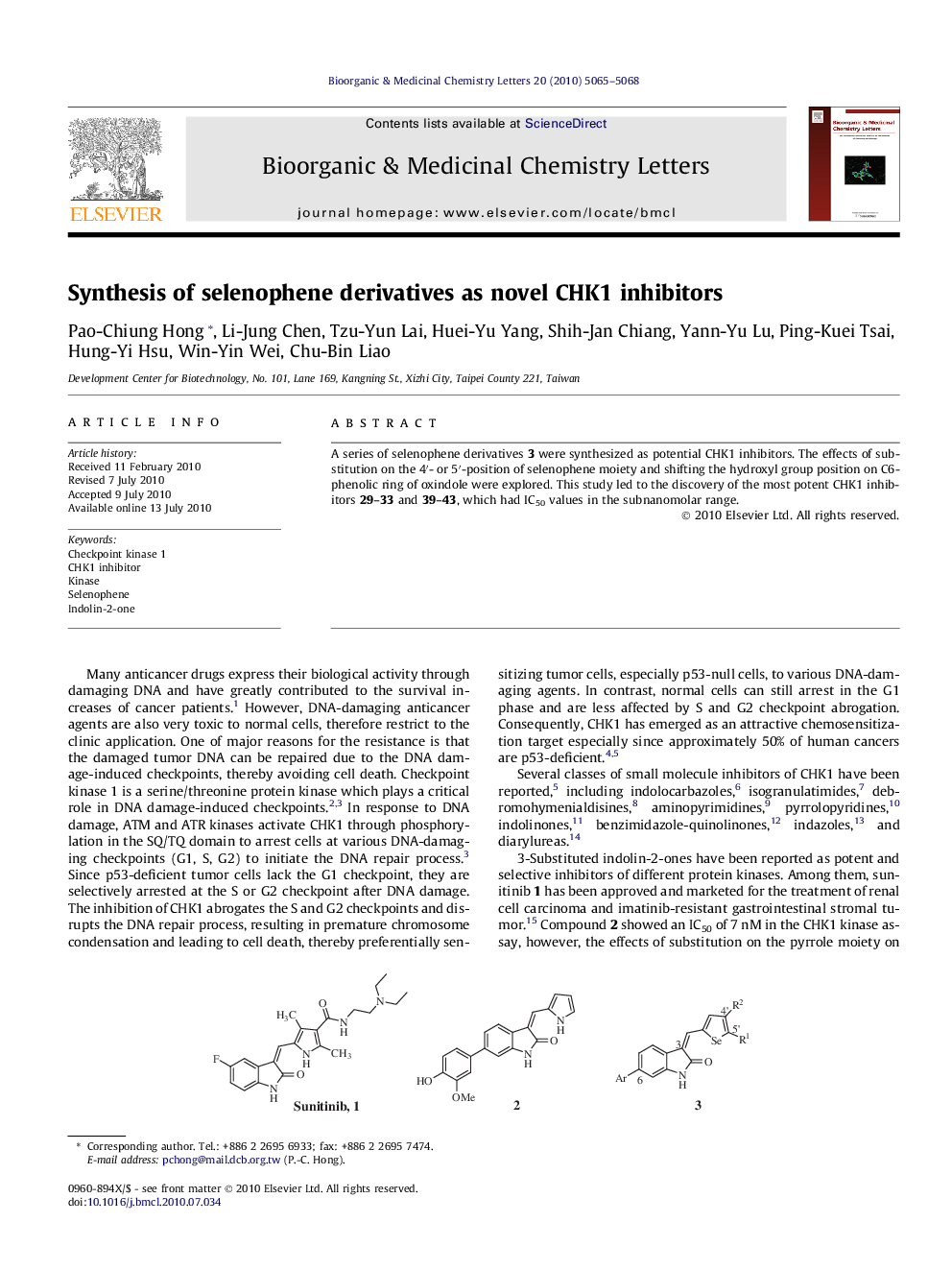
A series of selenophene derivatives 3 were synthesized as potential CHK1 inhibitors. The effects of substitution on the 4′- or 5′-position of selenophene moiety and shifting the hydroxyl group position on C6- phenolic ring of oxindole were explored. This study led to the discovery of the most potent CHK1 inhibitors 29–33 and 39–43, which had IC50 values in the subnanomolar range.
Graphical abstractA series of selenophene derivatives (i.e., 31) are reported as potent inhibitors of CHK1 kinase.Figure optionsDownload full-size imageDownload as PowerPoint slide
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Pao-Chiung Hong, Li-Jung Chen, Tzu-Yun Lai, Huei-Yu Yang, Shih-Jan Chiang, Yann-Yu Lu, Ping-Kuei Tsai, Hung-Yi Hsu, Win-Yin Wei, Chu-Bin Liao,