| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 1375966 | Bioorganic & Medicinal Chemistry Letters | 2009 | 4 Pages |
Abstract
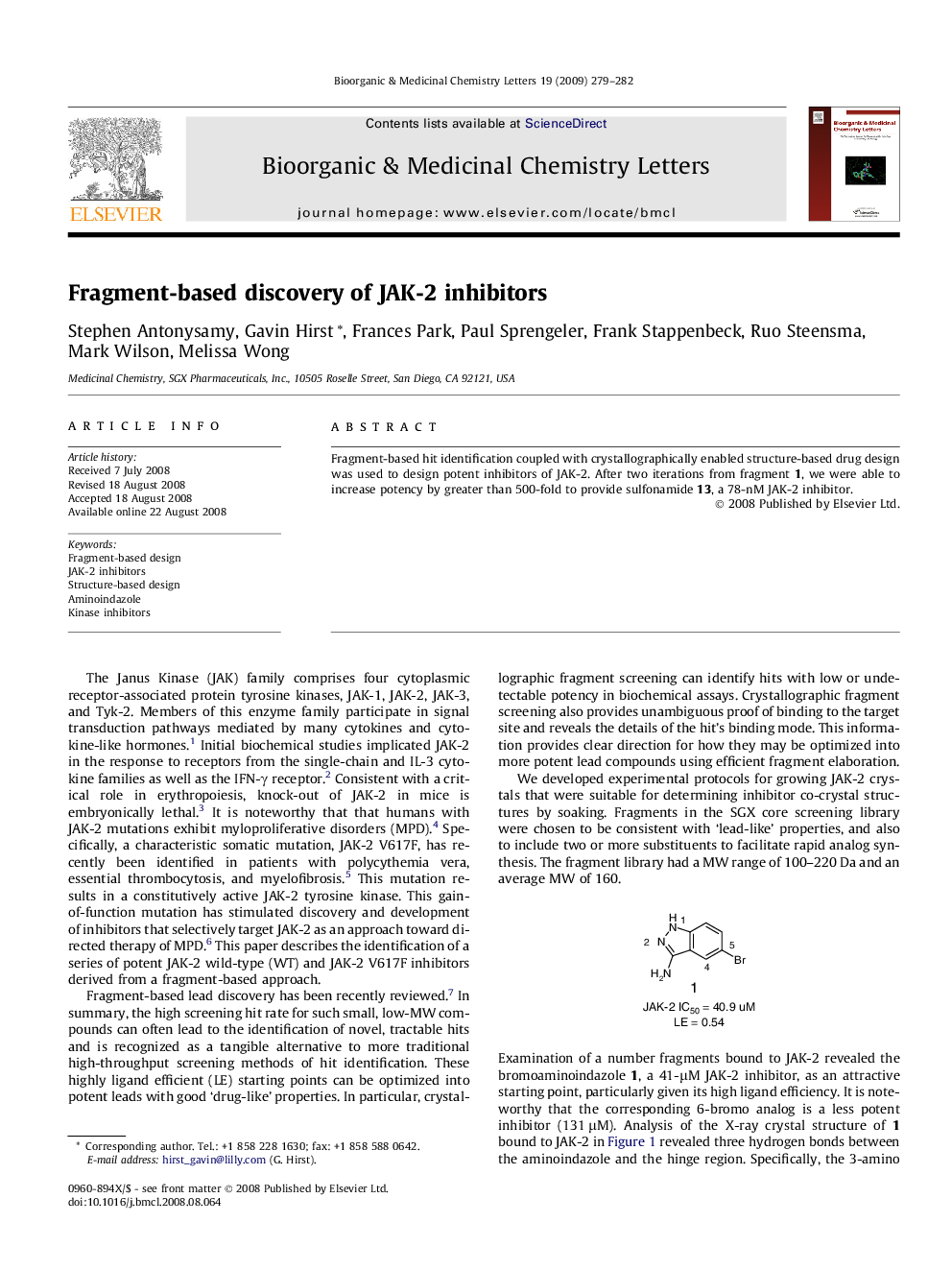
Fragment-based hit identification coupled with crystallographically enabled structure-based drug design was used to design potent inhibitors of JAK-2. After two iterations from fragment 1, we were able to increase potency by greater than 500-fold to provide sulfonamide 13, a 78-nM JAK-2 inhibitor.
Graphical abstractFragment-based hit identification coupled with crystallographically enabled structure-based drug design was used to design potent inhibitors of JAK-2. After two iterations from fragment 1, we were able to increase potency by greater than 500-fold to provide sulfonamide 13, a 78-nM JAK-2 inhibitor with greater than 35-fold selectivity versus JAK-3 (IC50 = 2.93 μM).Figure optionsDownload full-size imageDownload as PowerPoint slide
Related Topics
Physical Sciences and Engineering
Chemistry
Organic Chemistry
Authors
Stephen Antonysamy, Gavin Hirst, Frances Park, Paul Sprengeler, Frank Stappenbeck, Ruo Steensma, Mark Wilson, Melissa Wong,