| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 147285 | Chemical Engineering Journal | 2014 | 11 Pages |
•Synthetic hydroxyapatite as potential new material for CO2 capture at temperature higher than that of CaO based sorbents.•No appreciable decay of the absorbing activity upon several cycles.•The temperature of pre-treatment of porous granules impacts on sorption capacity.•CO2 carrying capacity very close to theoretical limit of 2.6% by mass.•Lower heat for regeneration in comparison with CaCO3 sorbents.
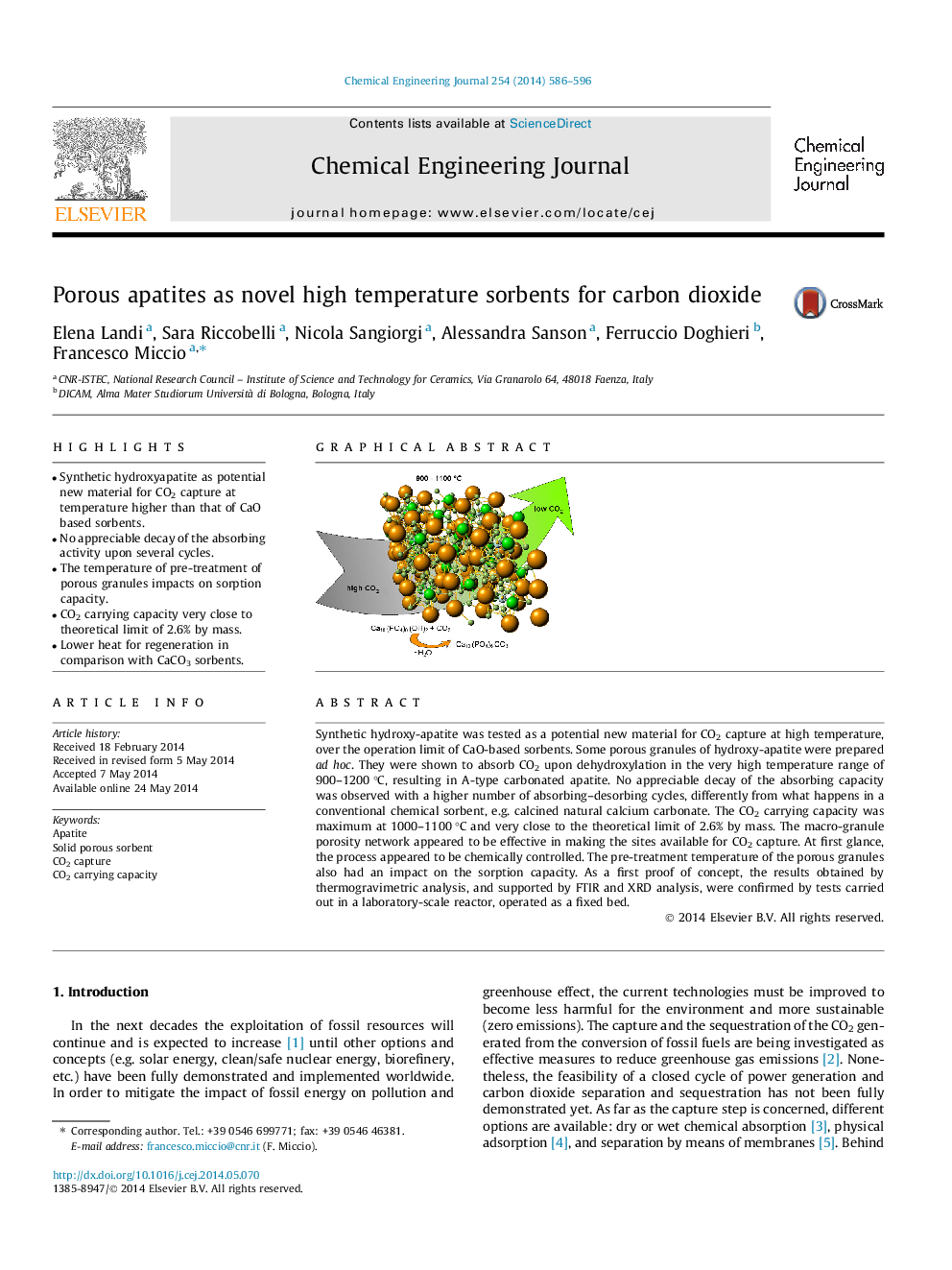
Synthetic hydroxy-apatite was tested as a potential new material for CO2 capture at high temperature, over the operation limit of CaO-based sorbents. Some porous granules of hydroxy-apatite were prepared ad hoc. They were shown to absorb CO2 upon dehydroxylation in the very high temperature range of 900–1200 °C, resulting in A-type carbonated apatite. No appreciable decay of the absorbing capacity was observed with a higher number of absorbing–desorbing cycles, differently from what happens in a conventional chemical sorbent, e.g. calcined natural calcium carbonate. The CO2 carrying capacity was maximum at 1000–1100 °C and very close to the theoretical limit of 2.6% by mass. The macro-granule porosity network appeared to be effective in making the sites available for CO2 capture. At first glance, the process appeared to be chemically controlled. The pre-treatment temperature of the porous granules also had an impact on the sorption capacity. As a first proof of concept, the results obtained by thermogravimetric analysis, and supported by FTIR and XRD analysis, were confirmed by tests carried out in a laboratory-scale reactor, operated as a fixed bed.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide