| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 148165 | Chemical Engineering Journal | 2013 | 9 Pages |
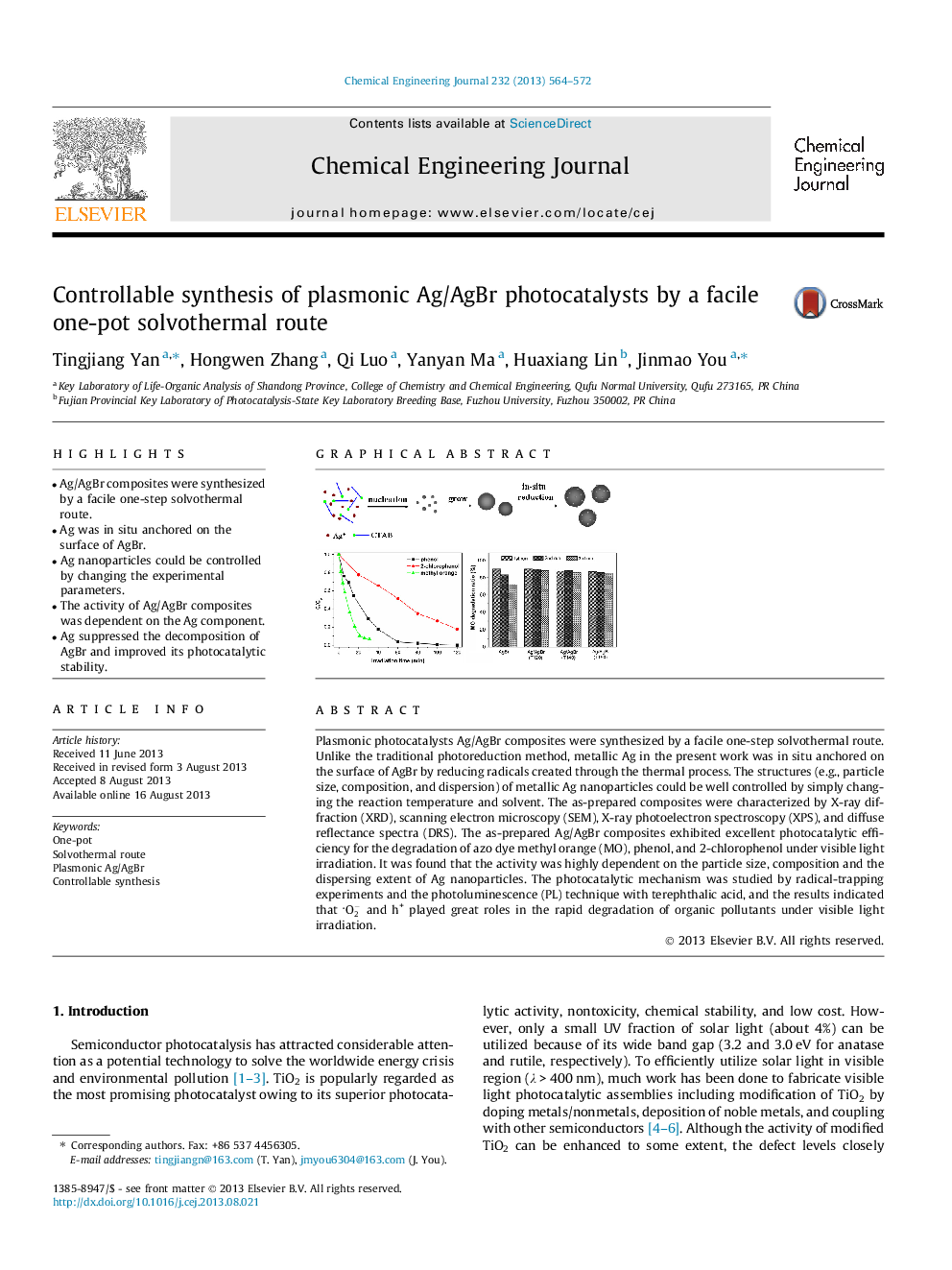
•Ag/AgBr composites were synthesized by a facile one-step solvothermal route.•Ag was in situ anchored on the surface of AgBr.•Ag nanoparticles could be controlled by changing the experimental parameters.•The activity of Ag/AgBr composites was dependent on the Ag component.•Ag suppressed the decomposition of AgBr and improved its photocatalytic stability.
Plasmonic photocatalysts Ag/AgBr composites were synthesized by a facile one-step solvothermal route. Unlike the traditional photoreduction method, metallic Ag in the present work was in situ anchored on the surface of AgBr by reducing radicals created through the thermal process. The structures (e.g., particle size, composition, and dispersion) of metallic Ag nanoparticles could be well controlled by simply changing the reaction temperature and solvent. The as-prepared composites were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS), and diffuse reflectance spectra (DRS). The as-prepared Ag/AgBr composites exhibited excellent photocatalytic efficiency for the degradation of azo dye methyl orange (MO), phenol, and 2-chlorophenol under visible light irradiation. It was found that the activity was highly dependent on the particle size, composition and the dispersing extent of Ag nanoparticles. The photocatalytic mechanism was studied by radical-trapping experiments and the photoluminescence (PL) technique with terephthalic acid, and the results indicated that O2- and h+ played great roles in the rapid degradation of organic pollutants under visible light irradiation.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slide