| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 148890 | Chemical Engineering Journal | 2013 | 9 Pages |
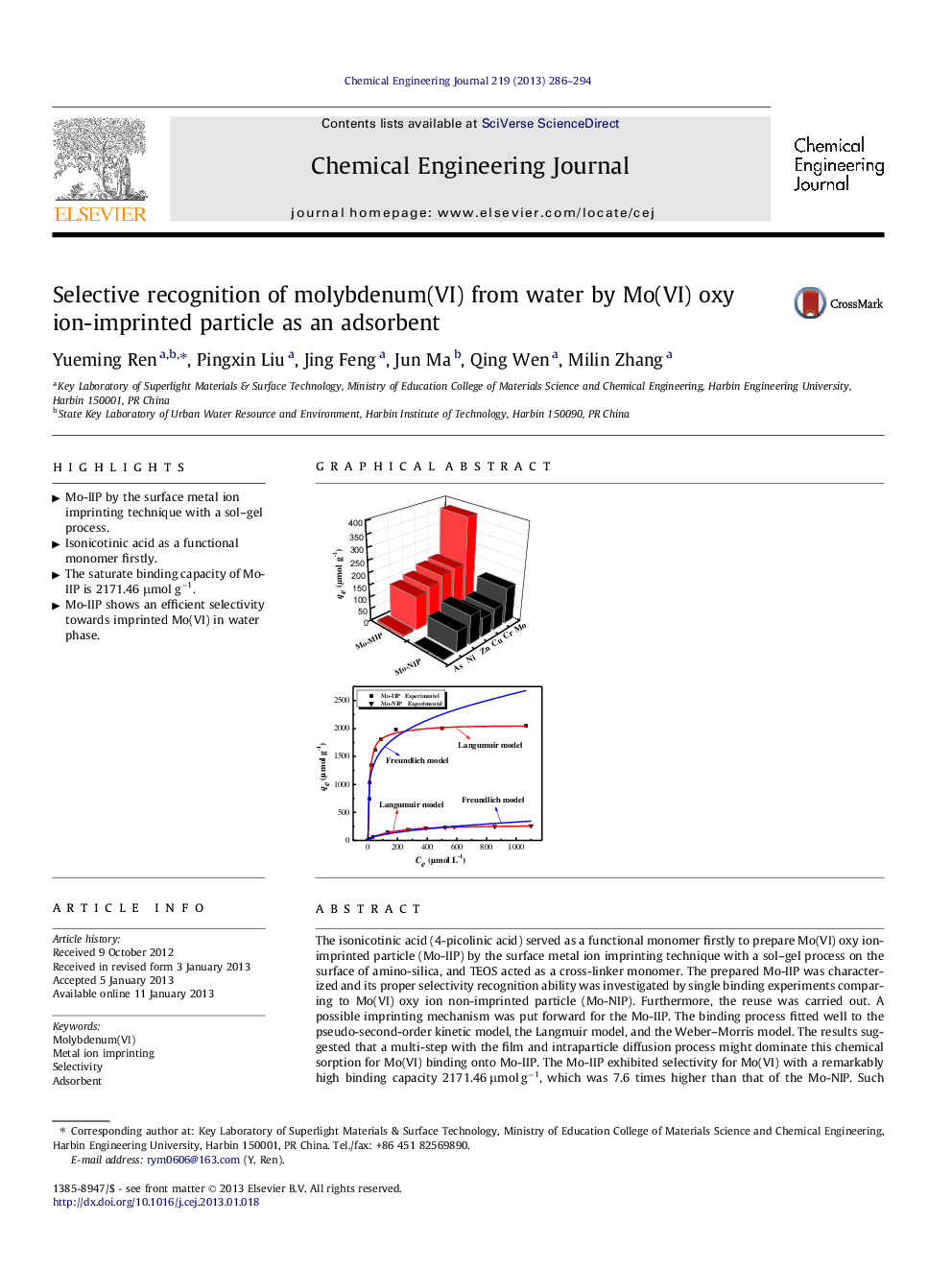
The isonicotinic acid (4-picolinic acid) served as a functional monomer firstly to prepare Mo(VI) oxy ion-imprinted particle (Mo-IIP) by the surface metal ion imprinting technique with a sol–gel process on the surface of amino-silica, and TEOS acted as a cross-linker monomer. The prepared Mo-IIP was characterized and its proper selectivity recognition ability was investigated by single binding experiments comparing to Mo(VI) oxy ion non-imprinted particle (Mo-NIP). Furthermore, the reuse was carried out. A possible imprinting mechanism was put forward for the Mo-IIP. The binding process fitted well to the pseudo-second-order kinetic model, the Langmuir model, and the Weber–Morris model. The results suggested that a multi-step with the film and intraparticle diffusion process might dominate this chemical sorption for Mo(VI) binding onto Mo-IIP. The Mo-IIP exhibited selectivity for Mo(VI) with a remarkably high binding capacity 2171.46 μmol g−1, which was 7.6 times higher than that of the Mo-NIP. Such efficient selectivity might be due to the complexation ability of nitrogen atom in the 4-picolinic acid with supper memory sites after imprinting process. The excellent affinity and exclusive selectivity make the Mo-IIP become an potential material for Mo(VI) removal and concentration from the water environment.
Graphical abstractFigure optionsDownload full-size imageDownload as PowerPoint slideHighlights► Mo-IIP by the surface metal ion imprinting technique with a sol–gel process. ► Isonicotinic acid as a functional monomer firstly. ► The saturate binding capacity of Mo-IIP is 2171.46 μmol g−1. ► Mo-IIP shows an efficient selectivity towards imprinted Mo(VI) in water phase.