| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 6587433 | Chemical Engineering Journal | 2013 | 14 Pages |
Abstract
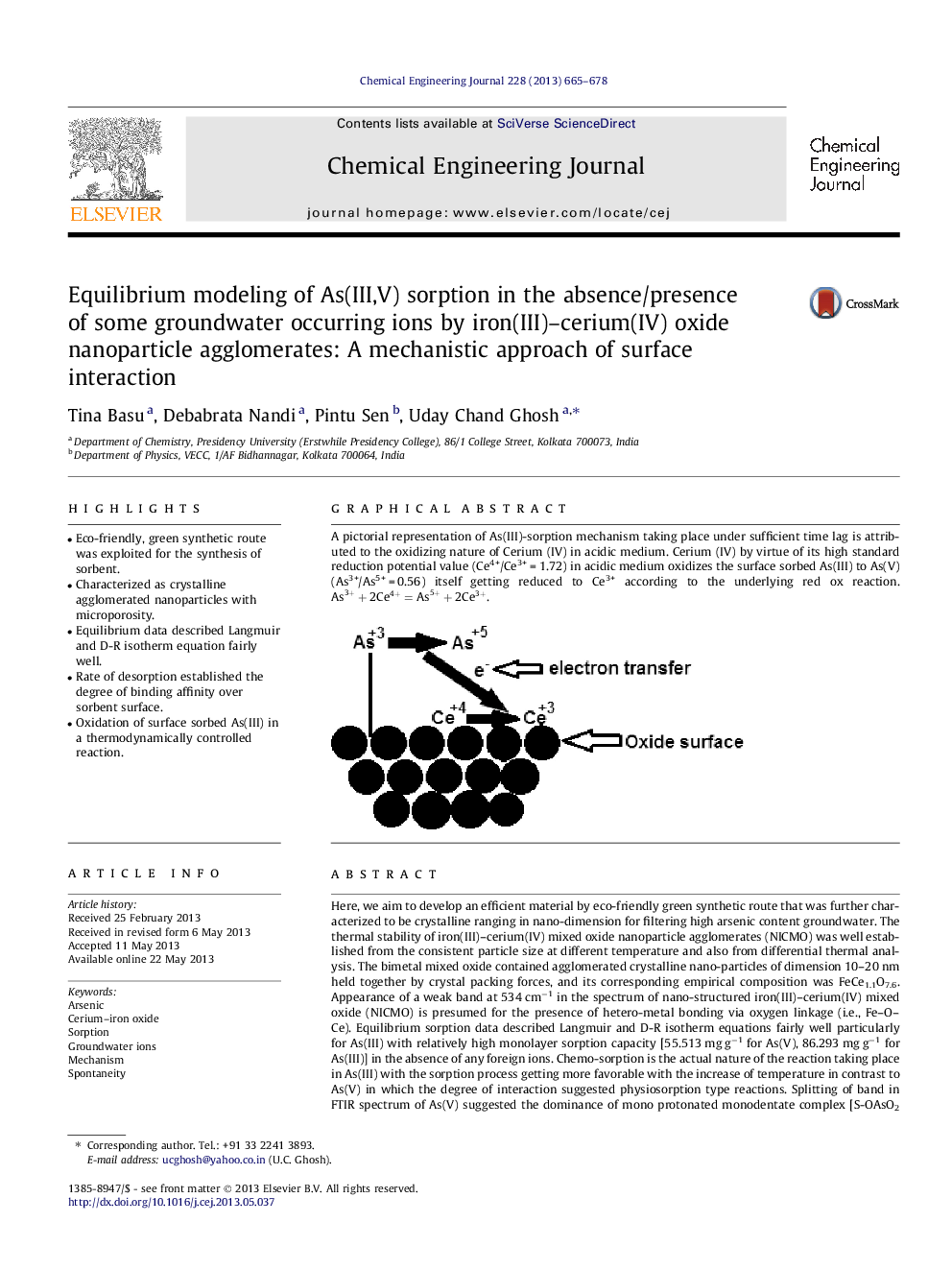
A pictorial representation of As(III)-sorption mechanism taking place under sufficient time lag is attributed to the oxidizing nature of Cerium (IV) in acidic medium. Cerium (IV) by virtue of its high standard reduction potential value (Ce4+/Ce3+Â =Â 1.72) in acidic medium oxidizes the surface sorbed As(III) to As(V) (As3+/As5+Â =Â 0.56) itself getting reduced to Ce3+ according to the underlying red ox reaction. As3++2Ce4+=As5++2Ce3+.
Keywords
Related Topics
Physical Sciences and Engineering
Chemical Engineering
Chemical Engineering (General)
Authors
Tina Basu, Debabrata Nandi, Pintu Sen, Uday Chand Ghosh,