| Article ID | Journal | Published Year | Pages | File Type |
|---|---|---|---|---|
| 2041718 | Cell Reports | 2016 | 15 Pages |
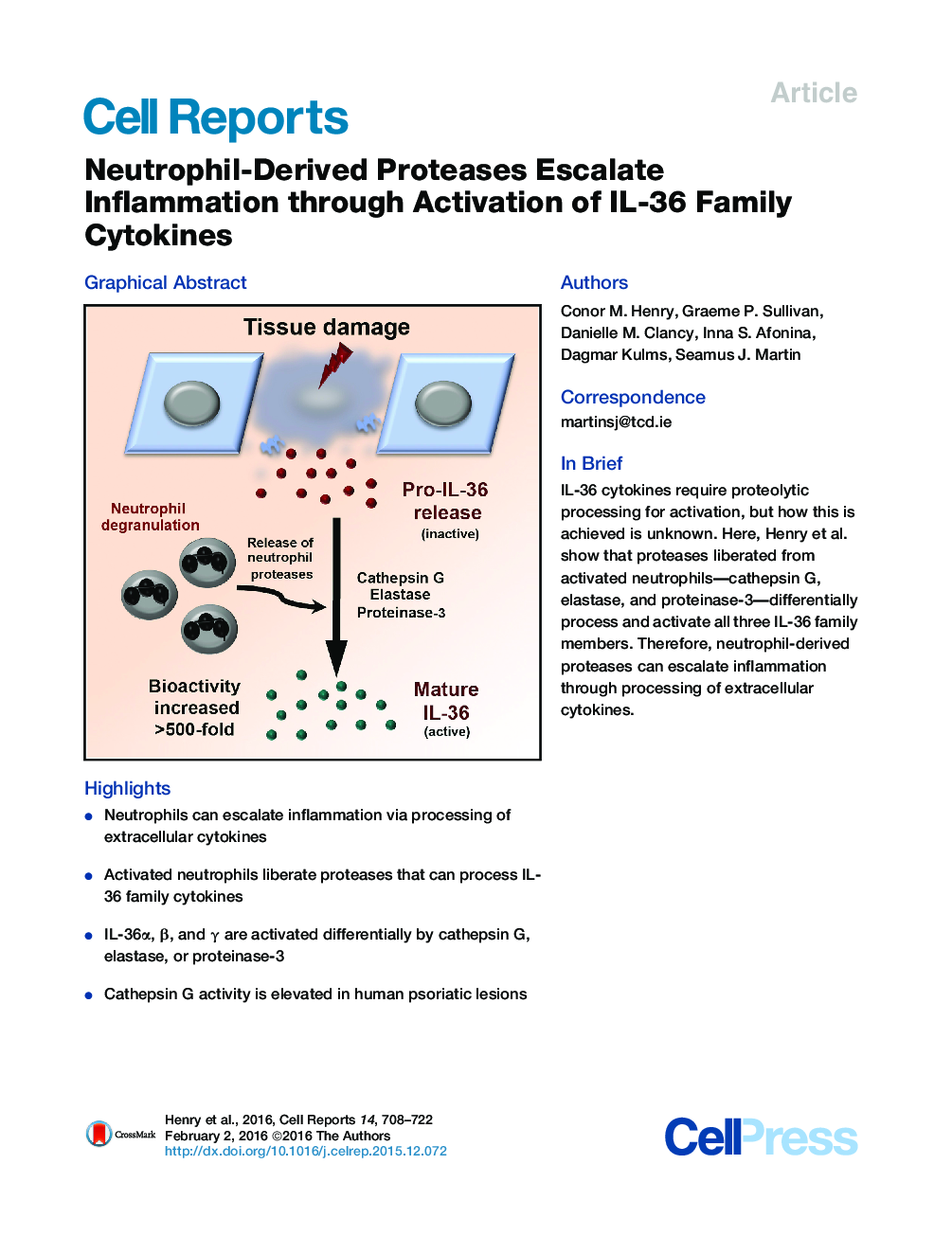
•Neutrophils can escalate inflammation via processing of extracellular cytokines•Activated neutrophils liberate proteases that can process IL-36 family cytokines•IL-36α, β, and γ are activated differentially by cathepsin G, elastase, or proteinase-3•Cathepsin G activity is elevated in human psoriatic lesions
SummaryRecent evidence has strongly implicated the IL-1 family cytokines IL-36α, IL-36β, and IL-36γ as key initiators of skin inflammation. Similar to the other members of the IL-1 family, IL-36 cytokines are expressed as inactive precursors and require proteolytic processing for activation; however, the responsible proteases are unknown. Here, we show that IL-36α, IL-36β, and IL-36γ are activated differentially by the neutrophil granule-derived proteases cathepsin G, elastase, and proteinase-3, increasing their biological activity ∼500-fold. Active IL-36 promoted a strong pro-inflammatory signature in primary keratinocytes and was sufficient to perturb skin differentiation in a reconstituted 3D human skin model, producing features resembling psoriasis. Furthermore, skin eluates from psoriasis patients displayed significantly elevated cathepsin G-like activity that was sufficient to activate IL-36β. These data identify neutrophil granule proteases as potent IL-36-activating enzymes, adding to our understanding of how neutrophils escalate inflammatory reactions. Inhibition of neutrophil-derived proteases may therefore have therapeutic benefits in psoriasis.
Graphical AbstractFigure optionsDownload full-size imageDownload as PowerPoint slide