| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229584 | 1495236 | 2014 | 7 صفحه PDF | دانلود رایگان |
• Three types of complexes were found for the complexes of hypohalous acids and phosphine derivatives.
• There is competition among hydrogen bond, halogen bond, and pnicogen bond.
• This competition is related to the nature of hypohalous acids and phosphine derivatives.
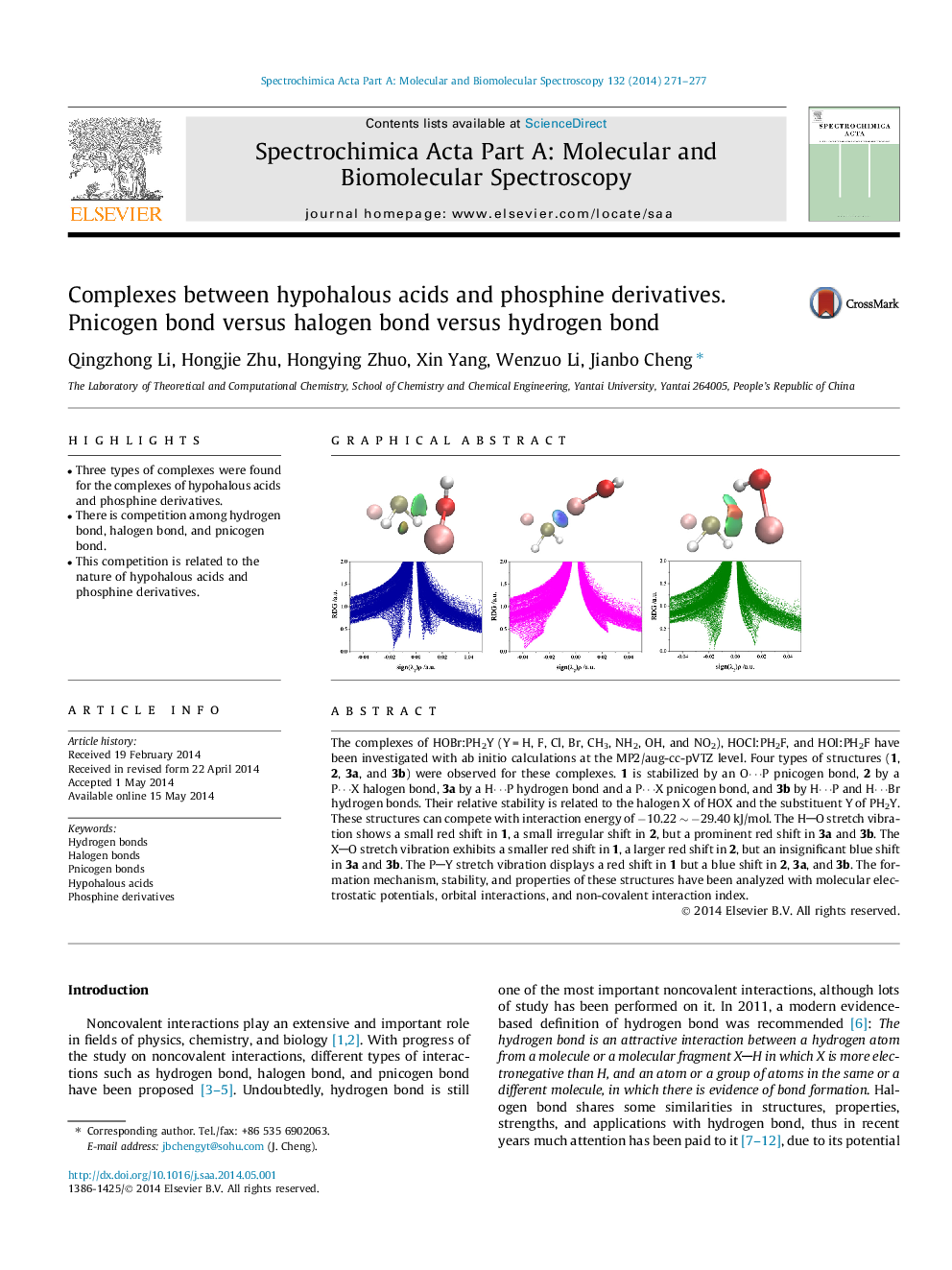
The complexes of HOBr:PH2Y (Y = H, F, Cl, Br, CH3, NH2, OH, and NO2), HOCl:PH2F, and HOI:PH2F have been investigated with ab initio calculations at the MP2/aug-cc-pVTZ level. Four types of structures (1, 2, 3a, and 3b) were observed for these complexes. 1 is stabilized by an O⋯P pnicogen bond, 2 by a P⋯X halogen bond, 3a by a H⋯P hydrogen bond and a P⋯X pnicogen bond, and 3b by H⋯P and H⋯Br hydrogen bonds. Their relative stability is related to the halogen X of HOX and the substituent Y of PH2Y. These structures can compete with interaction energy of −10.22 ∼ −29.40 kJ/mol. The HO stretch vibration shows a small red shift in 1, a small irregular shift in 2, but a prominent red shift in 3a and 3b. The XO stretch vibration exhibits a smaller red shift in 1, a larger red shift in 2, but an insignificant blue shift in 3a and 3b. The PY stretch vibration displays a red shift in 1 but a blue shift in 2, 3a, and 3b. The formation mechanism, stability, and properties of these structures have been analyzed with molecular electrostatic potentials, orbital interactions, and non-covalent interaction index.
Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 132, 11 November 2014, Pages 271–277