| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1356573 | 981130 | 2009 | 7 صفحه PDF | دانلود رایگان |
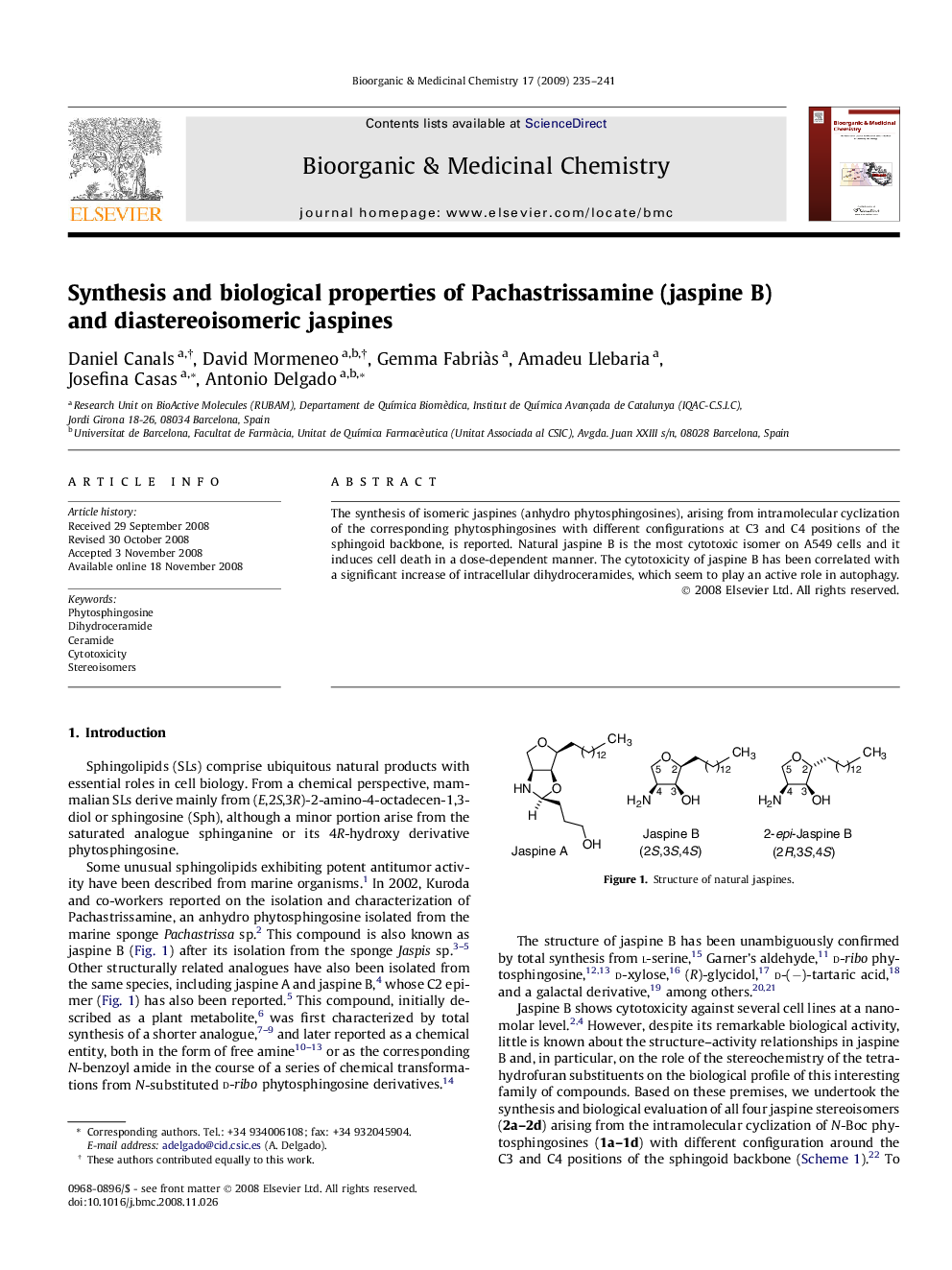
The synthesis of isomeric jaspines (anhydro phytosphingosines), arising from intramolecular cyclization of the corresponding phytosphingosines with different configurations at C3 and C4 positions of the sphingoid backbone, is reported. Natural jaspine B is the most cytotoxic isomer on A549 cells and it induces cell death in a dose-dependent manner. The cytotoxicity of jaspine B has been correlated with a significant increase of intracellular dihydroceramides, which seem to play an active role in autophagy.
The synthesis of jaspine B and three diastereomeric anhydro phytosphingosines is reported. Natural jaspine B was the most cytotoxic isomer on A549 cells and it induced cell death in a dose-dependent manner. The cytotoxicity of jaspine B has been correlated with a significant increase of intracellular dihydroceramides, which seem to play an active role in autophagy.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 17, Issue 1, 1 January 2009, Pages 235–241