| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1389550 | 982894 | 2009 | 6 صفحه PDF | دانلود رایگان |
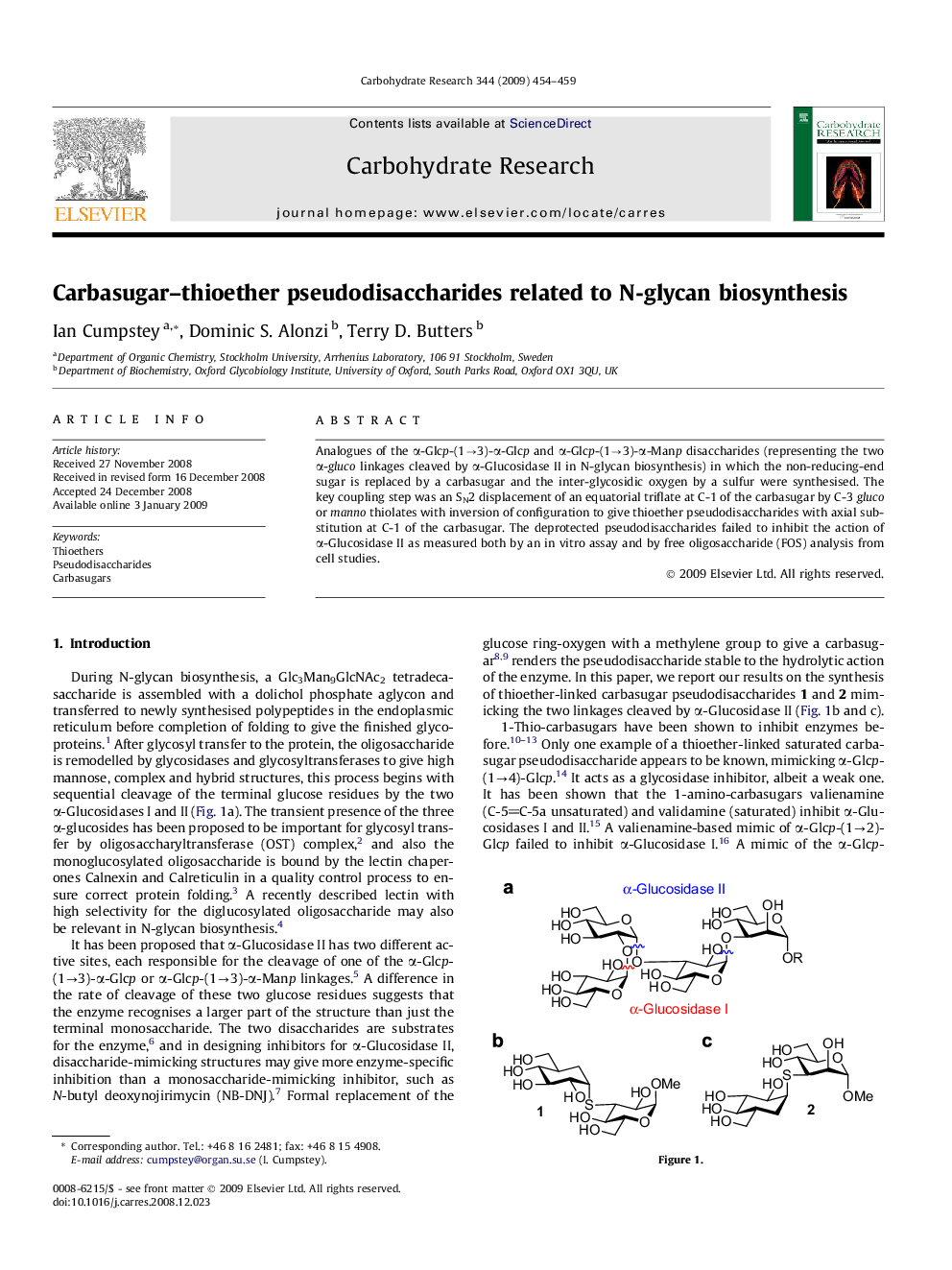
Analogues of the α-Glcp-(1→3)-α-Glcp and α-Glcp-(1→3)-α-Manp disaccharides (representing the two α-gluco linkages cleaved by α-Glucosidase II in N-glycan biosynthesis) in which the non-reducing-end sugar is replaced by a carbasugar and the inter-glycosidic oxygen by a sulfur were synthesised. The key coupling step was an SN2 displacement of an equatorial triflate at C-1 of the carbasugar by C-3 gluco or manno thiolates with inversion of configuration to give thioether pseudodisaccharides with axial substitution at C-1 of the carbasugar. The deprotected pseudodisaccharides failed to inhibit the action of α-Glucosidase II as measured both by an in vitro assay and by free oligosaccharide (FOS) analysis from cell studies.
Figure optionsDownload as PowerPoint slide
Journal: Carbohydrate Research - Volume 344, Issue 4, 10 March 2009, Pages 454–459