| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 4982506 | 1453861 | 2017 | 9 صفحه PDF | دانلود رایگان |
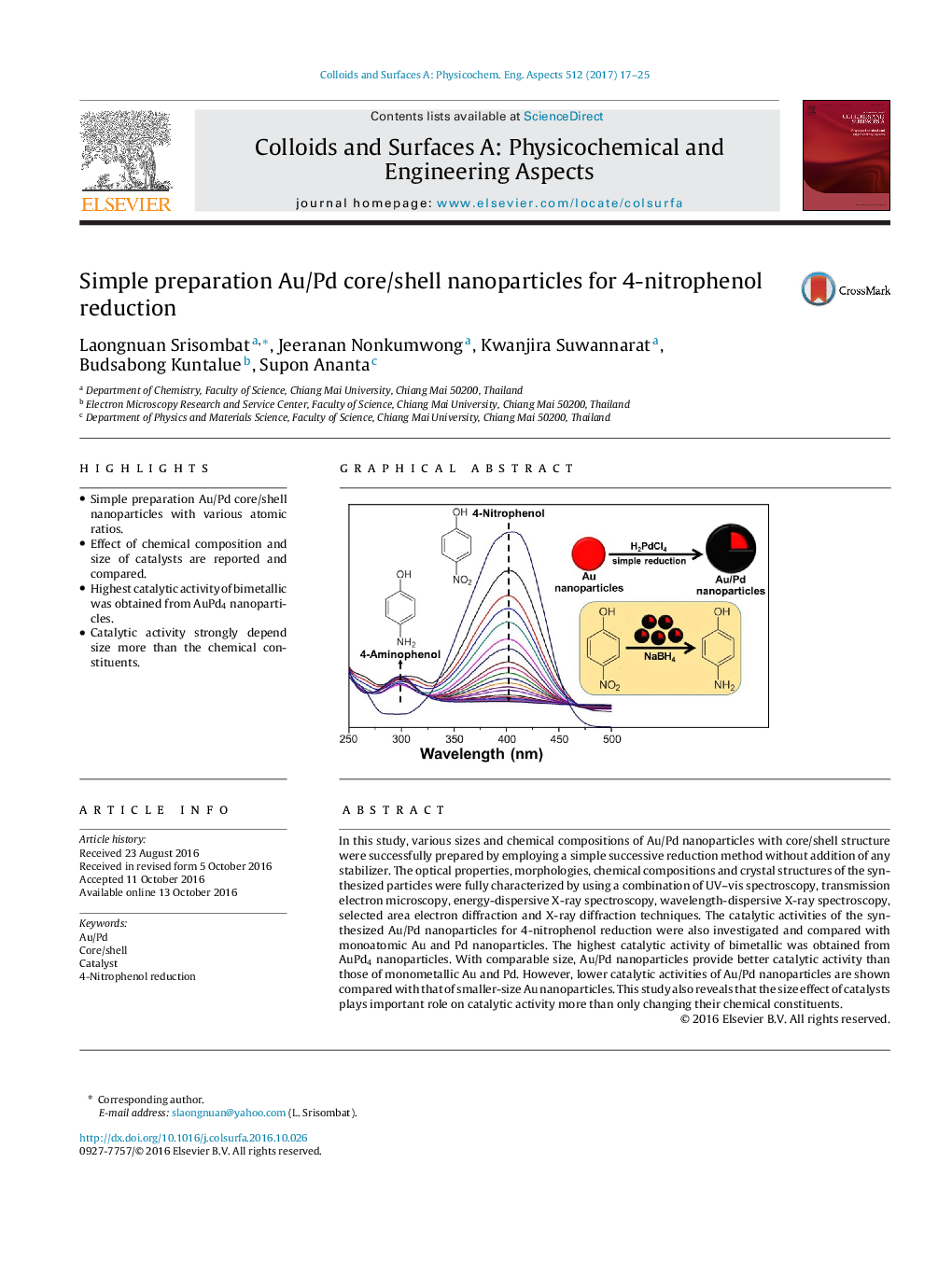
- Simple preparation Au/Pd core/shell nanoparticles with various atomic ratios.
- Effect of chemical composition and size of catalysts are reported and compared.
- Highest catalytic activity of bimetallic was obtained from AuPd4 nanoparticles.
- Catalytic activity strongly depend size more than the chemical constituents.
In this study, various sizes and chemical compositions of Au/Pd nanoparticles with core/shell structure were successfully prepared by employing a simple successive reduction method without addition of any stabilizer. The optical properties, morphologies, chemical compositions and crystal structures of the synthesized particles were fully characterized by using a combination of UV-vis spectroscopy, transmission electron microscopy, energy-dispersive X-ray spectroscopy, wavelength-dispersive X-ray spectroscopy, selected area electron diffraction and X-ray diffraction techniques. The catalytic activities of the synthesized Au/Pd nanoparticles for 4-nitrophenol reduction were also investigated and compared with monoatomic Au and Pd nanoparticles. The highest catalytic activity of bimetallic was obtained from AuPd4 nanoparticles. With comparable size, Au/Pd nanoparticles provide better catalytic activity than those of monometallic Au and Pd. However, lower catalytic activities of Au/Pd nanoparticles are shown compared with that of smaller-size Au nanoparticles. This study also reveals that the size effect of catalysts plays important role on catalytic activity more than only changing their chemical constituents.
186
Journal: Colloids and Surfaces A: Physicochemical and Engineering Aspects - Volume 512, 1 January 2017, Pages 17-25