| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 6466308 | 1422963 | 2017 | 10 صفحه PDF | دانلود رایگان |
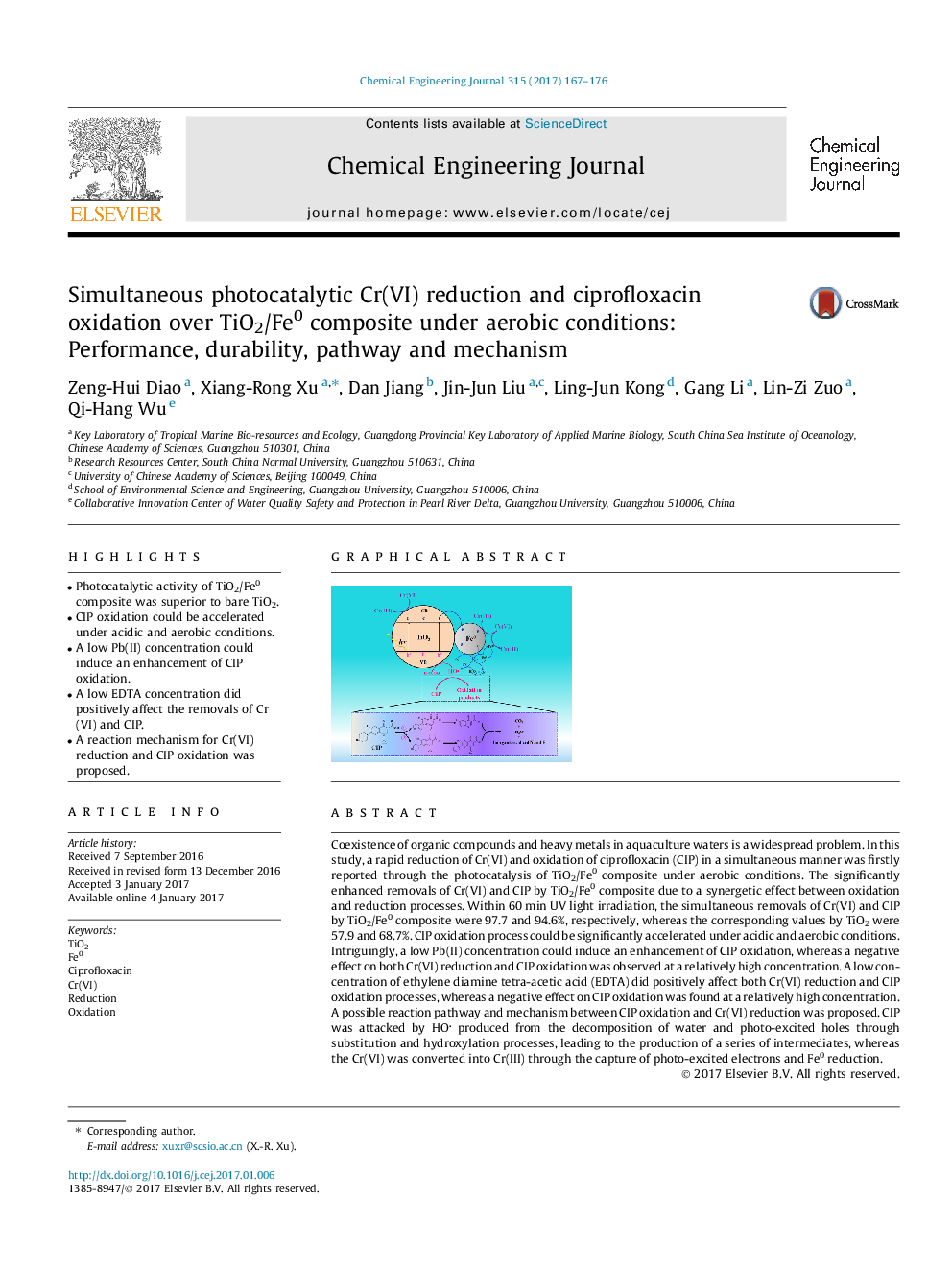
- Photocatalytic activity of TiO2/Fe0 composite was superior to bare TiO2.
- CIP oxidation could be accelerated under acidic and aerobic conditions.
- A low Pb(II) concentration could induce an enhancement of CIP oxidation.
- A low EDTA concentration did positively affect the removals of Cr(VI) and CIP.
- A reaction mechanism for Cr(VI) reduction and CIP oxidation was proposed.
Coexistence of organic compounds and heavy metals in aquaculture waters is a widespread problem. In this study, a rapid reduction of Cr(VI) and oxidation of ciprofloxacin (CIP) in a simultaneous manner was firstly reported through the photocatalysis of TiO2/Fe0 composite under aerobic conditions. The significantly enhanced removals of Cr(VI) and CIP by TiO2/Fe0 composite due to a synergetic effect between oxidation and reduction processes. Within 60Â min UV light irradiation, the simultaneous removals of Cr(VI) and CIP by TiO2/Fe0 composite were 97.7 and 94.6%, respectively, whereas the corresponding values by TiO2 were 57.9 and 68.7%. CIP oxidation process could be significantly accelerated under acidic and aerobic conditions. Intriguingly, a low Pb(II) concentration could induce an enhancement of CIP oxidation, whereas a negative effect on both Cr(VI) reduction and CIP oxidation was observed at a relatively high concentration. A low concentration of ethylene diamine tetra-acetic acid (EDTA) did positively affect both Cr(VI) reduction and CIP oxidation processes, whereas a negative effect on CIP oxidation was found at a relatively high concentration. A possible reaction pathway and mechanism between CIP oxidation and Cr(VI) reduction was proposed. CIP was attacked by HO produced from the decomposition of water and photo-excited holes through substitution and hydroxylation processes, leading to the production of a series of intermediates, whereas the Cr(VI) was converted into Cr(III) through the capture of photo-excited electrons and Fe0 reduction.
139
Journal: Chemical Engineering Journal - Volume 315, 1 May 2017, Pages 167-176