| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1404889 | 1501726 | 2015 | 7 صفحه PDF | دانلود رایگان |
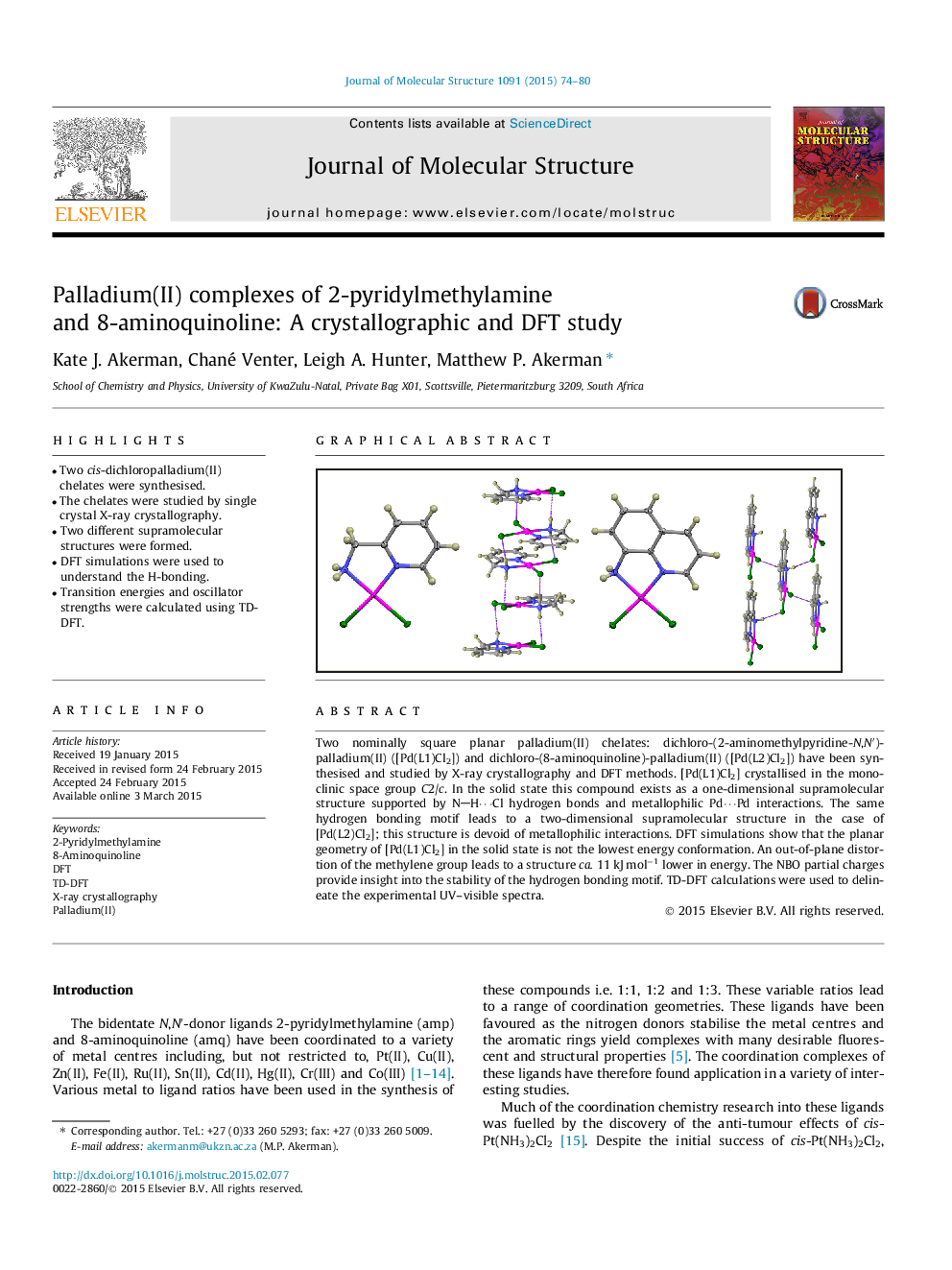
• Two cis-dichloropalladium(II) chelates were synthesised.
• The chelates were studied by single crystal X-ray crystallography.
• Two different supramolecular structures were formed.
• DFT simulations were used to understand the H-bonding.
• Transition energies and oscillator strengths were calculated using TD-DFT.
Two nominally square planar palladium(II) chelates: dichloro-(2-aminomethylpyridine-N,N′)-palladium(II) ([Pd(L1)Cl2]) and dichloro-(8-aminoquinoline)-palladium(II) ([Pd(L2)Cl2]) have been synthesised and studied by X-ray crystallography and DFT methods. [Pd(L1)Cl2] crystallised in the monoclinic space group C2/c. In the solid state this compound exists as a one-dimensional supramolecular structure supported by NH⋯Cl hydrogen bonds and metallophilic Pd⋯Pd interactions. The same hydrogen bonding motif leads to a two-dimensional supramolecular structure in the case of [Pd(L2)Cl2]; this structure is devoid of metallophilic interactions. DFT simulations show that the planar geometry of [Pd(L1)Cl2] in the solid state is not the lowest energy conformation. An out-of-plane distortion of the methylene group leads to a structure ca. 11 kJ mol−1 lower in energy. The NBO partial charges provide insight into the stability of the hydrogen bonding motif. TD-DFT calculations were used to delineate the experimental UV–visible spectra.
Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volume 1091, 5 July 2015, Pages 74–80