| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1344314 | 1500361 | 2012 | 4 صفحه PDF | دانلود رایگان |
عنوان انگلیسی مقاله ISI
From carnitinamide to 5-aminomethyl-2-oxazolidinones
دانلود مقاله + سفارش ترجمه
دانلود مقاله ISI انگلیسی
رایگان برای ایرانیان
موضوعات مرتبط
مهندسی و علوم پایه
شیمی
شیمی معدنی
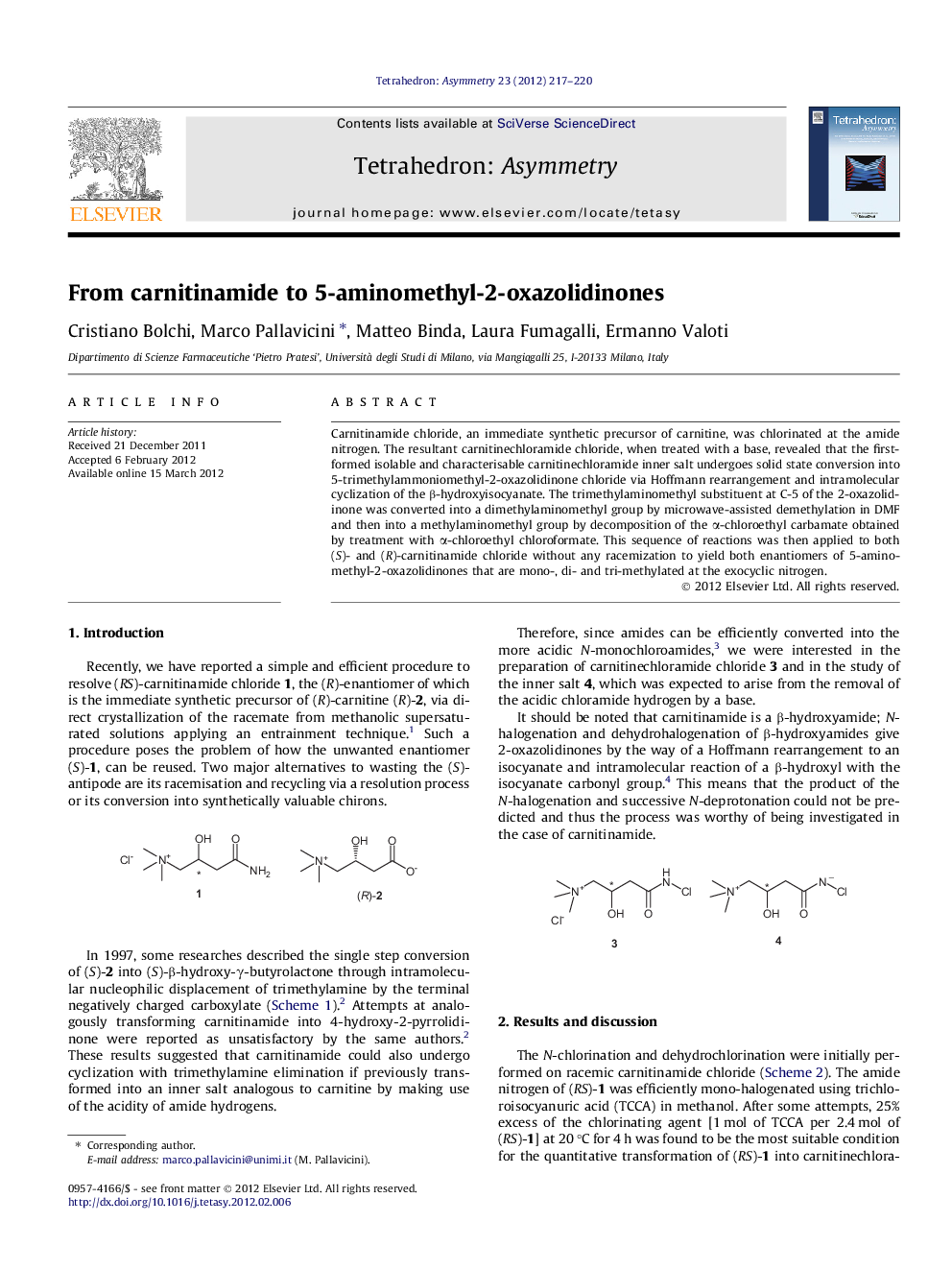
پیش نمایش صفحه اول مقاله
چکیده انگلیسی
Carnitinamide chloride, an immediate synthetic precursor of carnitine, was chlorinated at the amide nitrogen. The resultant carnitinechloramide chloride, when treated with a base, revealed that the first-formed isolable and characterisable carnitinechloramide inner salt undergoes solid state conversion into 5-trimethylammoniomethyl-2-oxazolidinone chloride via Hoffmann rearrangement and intramolecular cyclization of the β-hydroxyisocyanate. The trimethylaminomethyl substituent at C-5 of the 2-oxazolidinone was converted into a dimethylaminomethyl group by microwave-assisted demethylation in DMF and then into a methylaminomethyl group by decomposition of the α-chloroethyl carbamate obtained by treatment with α-chloroethyl chloroformate. This sequence of reactions was then applied to both (S)- and (R)-carnitinamide chloride without any racemization to yield both enantiomers of 5-aminomethyl-2-oxazolidinones that are mono-, di- and tri-methylated at the exocyclic nitrogen.
ناشر
Database: Elsevier - ScienceDirect (ساینس دایرکت)
Journal: Tetrahedron: Asymmetry - Volume 23, Issues 3â4, 29 February 2012, Pages 217-220
Journal: Tetrahedron: Asymmetry - Volume 23, Issues 3â4, 29 February 2012, Pages 217-220
نویسندگان
Cristiano Bolchi, Marco Pallavicini, Matteo Binda, Laura Fumagalli, Ermanno Valoti,