| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1345494 | 980204 | 2007 | 8 صفحه PDF | دانلود رایگان |
عنوان انگلیسی مقاله ISI
Synthesis of conformationally restricted acetylcholine analogues. Comparing lipase-mediated resolution with simulated moving bed chromatography of arylated β-hydroxy-pyrrolidines
دانلود مقاله + سفارش ترجمه
دانلود مقاله ISI انگلیسی
رایگان برای ایرانیان
موضوعات مرتبط
مهندسی و علوم پایه
شیمی
شیمی معدنی
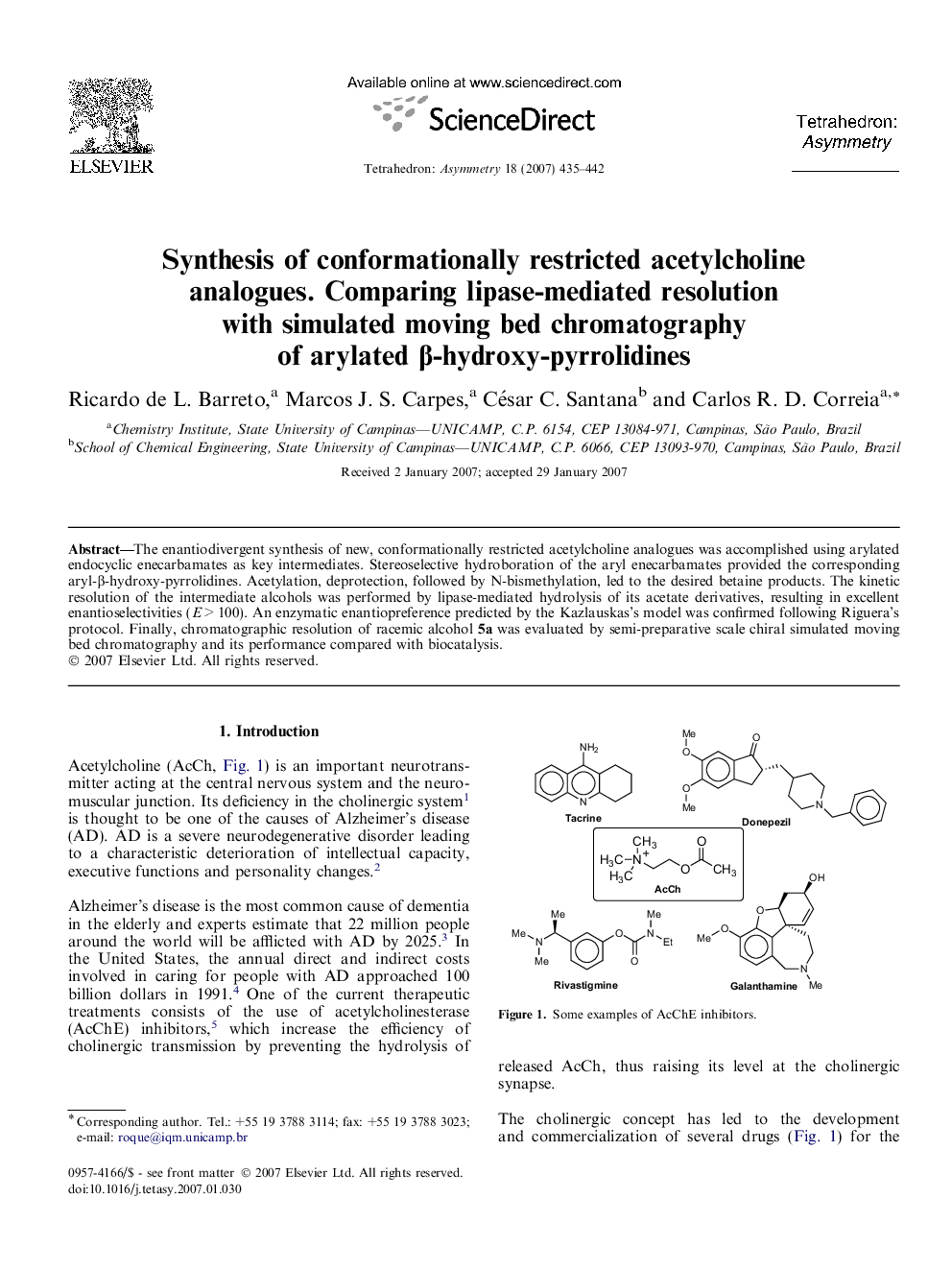
پیش نمایش صفحه اول مقاله
چکیده انگلیسی
The enantiodivergent synthesis of new, conformationally restricted acetylcholine analogues was accomplished using arylated endocyclic enecarbamates as key intermediates. Stereoselective hydroboration of the aryl enecarbamates provided the corresponding aryl-β-hydroxy-pyrrolidines. Acetylation, deprotection, followed by N-bismethylation, led to the desired betaine products. The kinetic resolution of the intermediate alcohols was performed by lipase-mediated hydrolysis of its acetate derivatives, resulting in excellent enantioselectivities (E > 100). An enzymatic enantiopreference predicted by the Kazlauskas's model was confirmed following Riguera's protocol. Finally, chromatographic resolution of racemic alcohol 5a was evaluated by semi-preparative scale chiral simulated moving bed chromatography and its performance compared with biocatalysis.
ناشر
Database: Elsevier - ScienceDirect (ساینس دایرکت)
Journal: Tetrahedron: Asymmetry - Volume 18, Issue 3, 28 February 2007, Pages 435-442
Journal: Tetrahedron: Asymmetry - Volume 18, Issue 3, 28 February 2007, Pages 435-442
نویسندگان
Ricardo de L. Barreto, Marcos J.S. Carpes, César C. Santana, Carlos R.D. Correia,