| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1347956 | 980332 | 2008 | 5 صفحه PDF | دانلود رایگان |
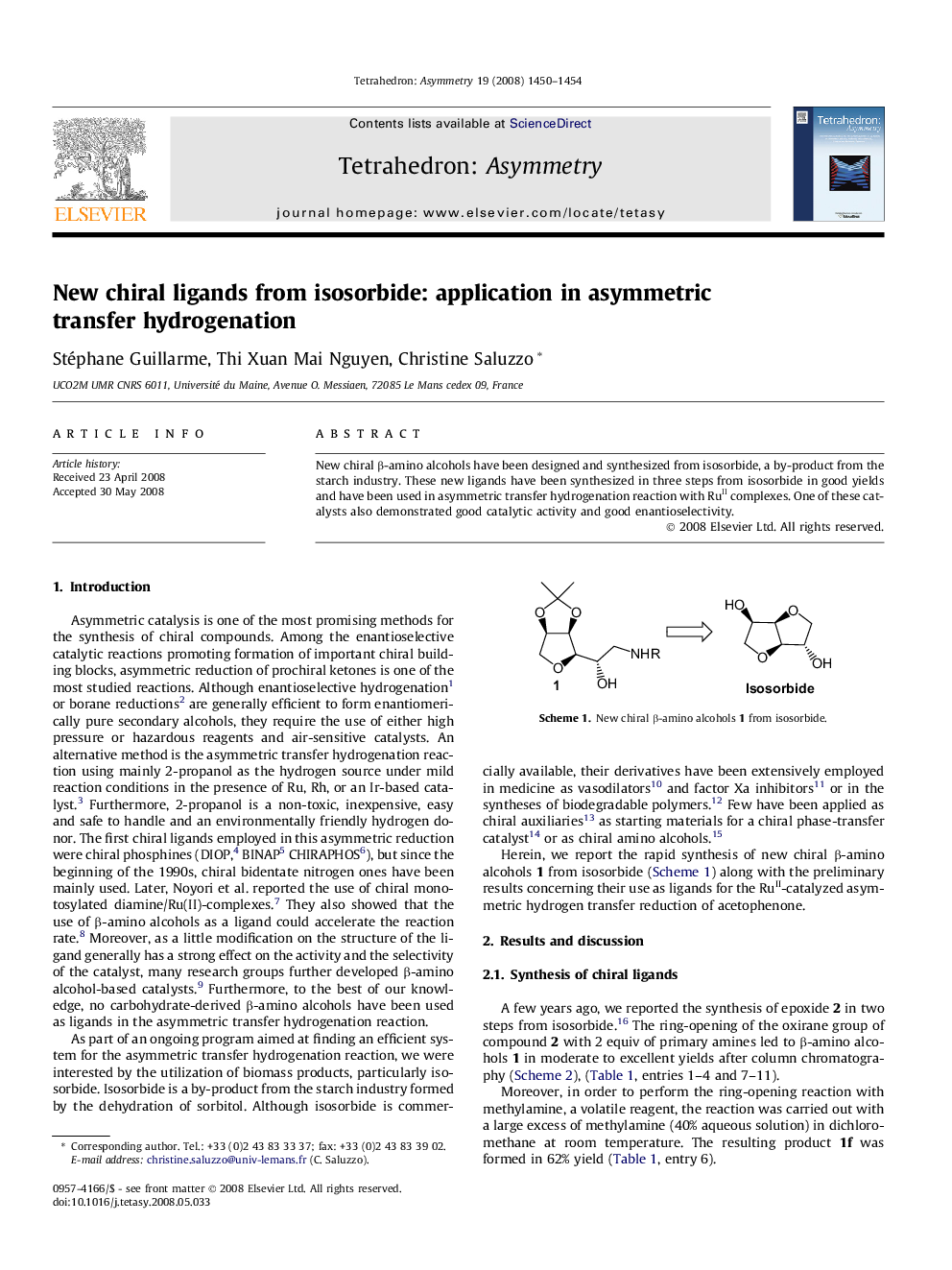
New chiral β-amino alcohols have been designed and synthesized from isosorbide, a by-product from the starch industry. These new ligands have been synthesized in three steps from isosorbide in good yields and have been used in asymmetric transfer hydrogenation reaction with RuII complexes. One of these catalysts also demonstrated good catalytic activity and good enantioselectivity.
Figure optionsDownload as PowerPoint slide
3,6-Anhydro-1-(benzylamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC16H23NO4[α]D25=-57.5 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-[(S)-(1-hydroxybutan-2-ylamino)]-1-deoxy-4,5-O-isopropylidene-d-sorbitolC13H25NO5[α]D25=-35.3 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (1′S,2S,3R,4R,5R)
3,6-Anhydro-1-(methylamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC10H19NO4[α]D25=-54.2 (c 0.5, CH3OH)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-(isopropylamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC12H23NO4[α]D25=-58.8 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-(cyclohexylamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC15H27NO4[α]D25=-58.2 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-[(R)-α-méthylbenzylamino)]-1-deoxy-4,5-O-isopropylidene-d-sorbitolC17H25NO4[α]D25=-17.3 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (1′R,2S,3R,4R,5R)
3,6-Anhydro-1-(benzeneamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC12H23NO4[α]D25=-70.0 (c 0.25, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-(amino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC12H23NO4[α]D25=-40.0 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-(tertiobutylamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC13H25NO4[α]D25=-62.3 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-((R)-(1-hydroxybutan-2-ylamino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC13H25NO5[α]D25=-64.0 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (1′R,2S,3R,4R,5R)
3,6-Anhydro-1-(morpholino)-1-deoxy-4,5-O-isopropylidene-d-sorbitolC13H23NO5[α]D25=-63.8 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (2S,3R,4R,5R)
3,6-Anhydro-1-[(S)-α-méthylbenzylamino)]-1-deoxy-4,5-O-isopropylidene-d-sorbitolC17H25NO4[α]D25=-90.0 (c 1.0, CH2Cl2)Source of chirality: isosorbideAbsolute configuration: (1′S,2S,3R,4R,5R)
Journal: Tetrahedron: Asymmetry - Volume 19, Issue 12, 30 June 2008, Pages 1450–1454