| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1370375 | 981816 | 2013 | 5 صفحه PDF | دانلود رایگان |
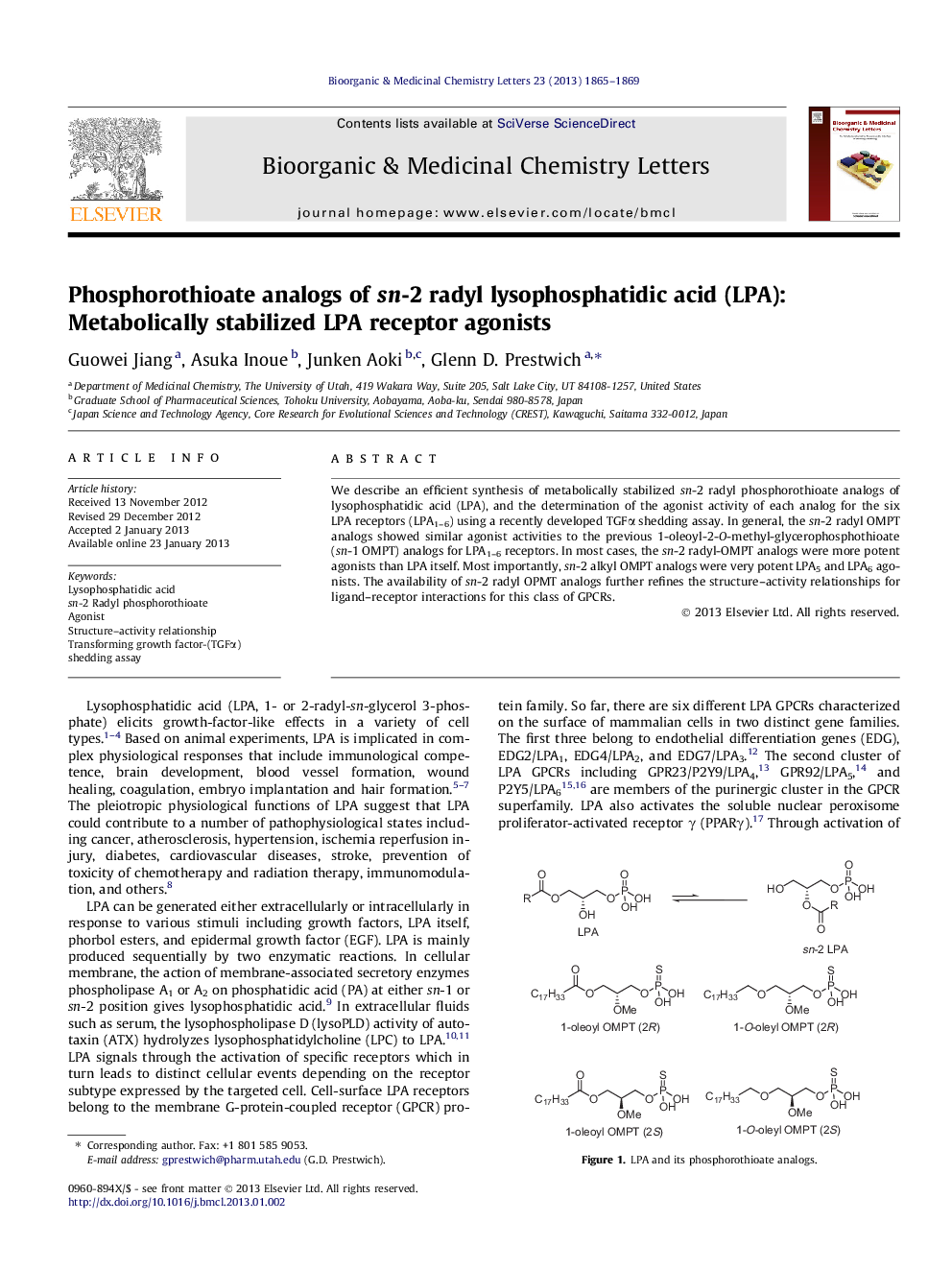
We describe an efficient synthesis of metabolically stabilized sn-2 radyl phosphorothioate analogs of lysophosphatidic acid (LPA), and the determination of the agonist activity of each analog for the six LPA receptors (LPA1–6) using a recently developed TGFα shedding assay. In general, the sn-2 radyl OMPT analogs showed similar agonist activities to the previous 1-oleoyl-2-O-methyl-glycerophosphothioate (sn-1 OMPT) analogs for LPA1–6 receptors. In most cases, the sn-2 radyl-OMPT analogs were more potent agonists than LPA itself. Most importantly, sn-2 alkyl OMPT analogs were very potent LPA5 and LPA6 agonists. The availability of sn-2 radyl OPMT analogs further refines the structure–activity relationships for ligand–receptor interactions for this class of GPCRs.
Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry Letters - Volume 23, Issue 6, 15 March 2013, Pages 1865–1869