| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1343962 | 980052 | 2013 | 5 صفحه PDF | دانلود رایگان |
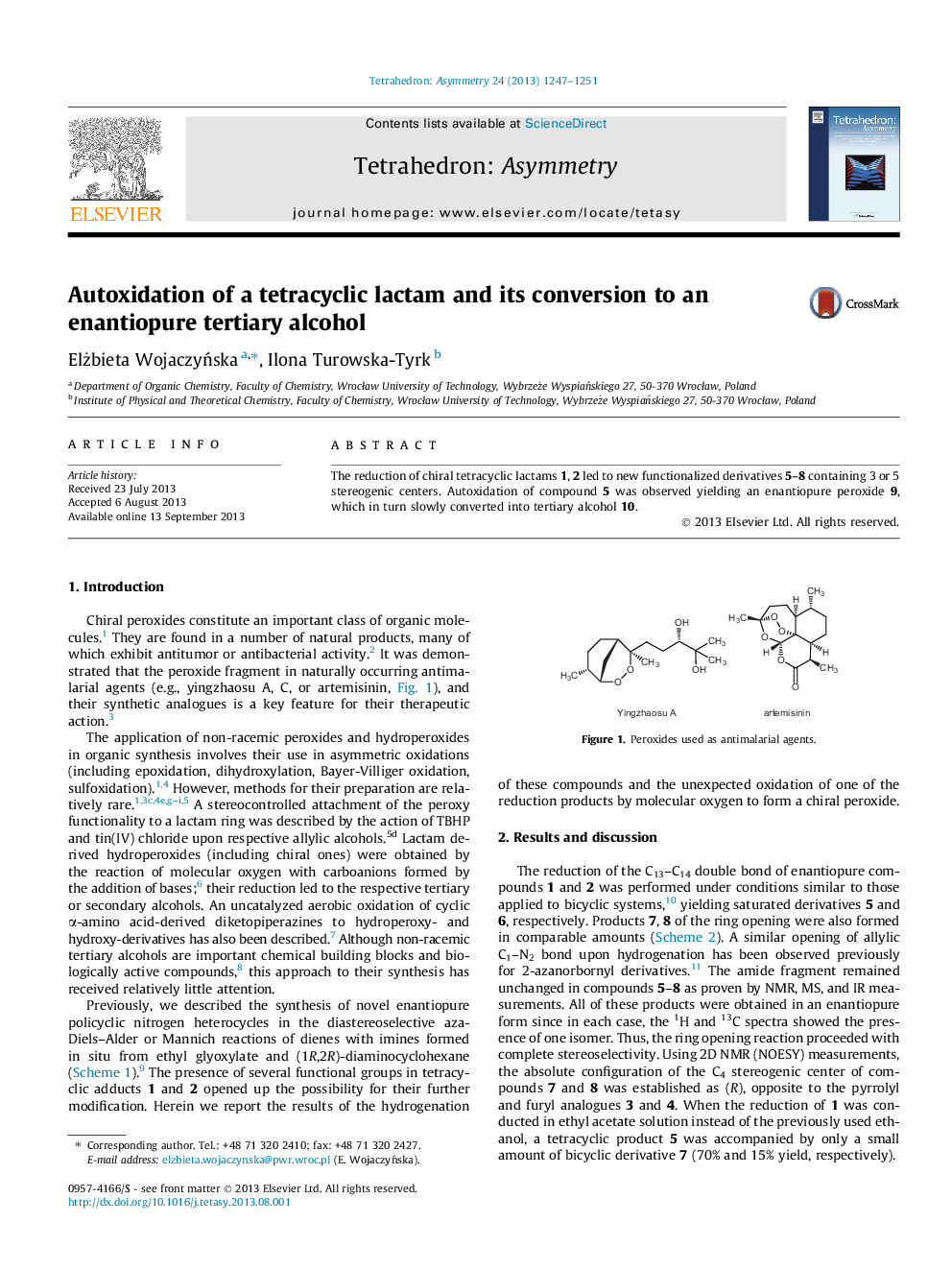
The reduction of chiral tetracyclic lactams 1, 2 led to new functionalized derivatives 5–8 containing 3 or 5 stereogenic centers. Autoxidation of compound 5 was observed yielding an enantiopure peroxide 9, which in turn slowly converted into tertiary alcohol 10.
Figure optionsDownload as PowerPoint slide
(1R,3R,8R,11R,12S)-10-Oxo-2,9-diazatetracyclo[10.2.1.02.11.03.8]pentadecaneC13H20N2O[α]D20=+98.2 (c 1.12, CH2Cl2)Source of chirality: chiral substrateAbsolute configuration: (1R,3R,8R,11R,12S) (by chemical correlation)
(3R,8R,11R)-10-Oxo-2,9-diazatetracyclo[10.2.2.02.11.03.8]hexadecaneC14H22N2O[α]D20=+18.0 (c 0.25, CH2Cl2)Source of chirality: chiral substrateAbsolute configuration: (3R,8R,11R) (by chemical correlation)
(1R,4R,6R)-4-Cyclopentyl-3-oxo-2,5-diazabicyclo[4.4.0]decaneC13H22N2O[α]D20=+31.0 (c 0.55, CH2Cl2)Source of chirality: chiral substrateAbsolute configuration: (1R,4R,6R) (by chemical correlation and 2D NMR NOESY measurement)
(1R,4R,6R)-4-Cyclohexyl-3-oxo-2,5-diazabicyclo[4.4.0]decaneC14H24N2O[α]D20=+20.7 (c 0.53, CH2Cl2)Source of chirality: chiral substrateAbsolute configuration: (1R,4R,6R) (by chemical correlation and 2D NMR NOESY measurement)
Bis-11,11′-{(1R,3R,8R,11S,12S)-10-oxo-2,9-diazatetracyclo[10.2.1.02.11.03.8]pentadecyl}peroxideC26H38N4O4[α]D20=+58.3 (c 0.57, CH2Cl2)Source of chirality: chiral substrateAbsolute configuration: (1R,3R,8R,11S,12S) (by chemical correlation and X-ray measurement)
(1R,3R,8R,11S,12S)-11-Hydroxy-10-oxo-2,9-diazatetracyclo[10.2.1.02.11.03.8]pentadecaneC13H20N2O2[α]D20=+75.1 (c 0.75, CH2Cl2)Source of chirality: chiral substrateAbsolute configuration: (1R,3R,8R,11S,12S) (by chemical correlation and X-ray measurement)
Journal: Tetrahedron: Asymmetry - Volume 24, Issue 19, 15 October 2013, Pages 1247–1251