| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1344258 | 980083 | 2012 | 5 صفحه PDF | دانلود رایگان |
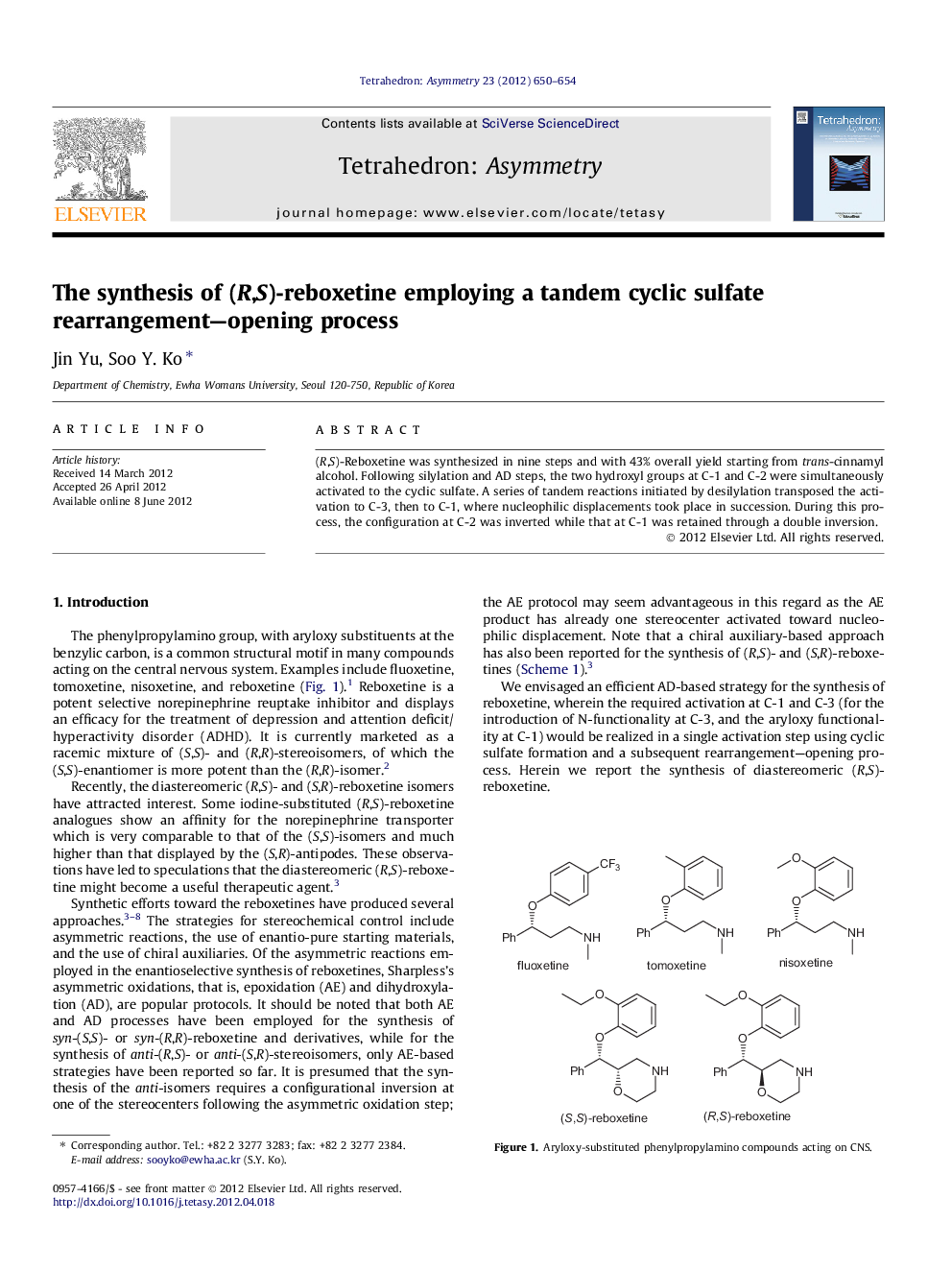
(R,S)-Reboxetine was synthesized in nine steps and with 43% overall yield starting from trans-cinnamyl alcohol. Following silylation and AD steps, the two hydroxyl groups at C-1 and C-2 were simultaneously activated to the cyclic sulfate. A series of tandem reactions initiated by desilylation transposed the activation to C-3, then to C-1, where nucleophilic displacements took place in succession. During this process, the configuration at C-2 was inverted while that at C-1 was retained through a double inversion.
Figure optionsDownload as PowerPoint slide
(1S,2R)-3-Azido-1-(2-ethoxyphenoxy)-1-phenylpropan-2-olC17H19N3O3[α]D29=+33.8(c1.04,EtOH)Absolute configuration: (1S,2R)Source of chirality: asymmetric synthesis
(R)-2-[(S)-(2-Ethoxyphenoxy)(phenyl)methyl]morpholine [(R,S)-reboxetine]C19H23NO3[α]D28=+16.0(c0.68,CH2Cl2)Absolute configuration: (2R,1’S)Source of chirality: asymmetric synthesis
Journal: Tetrahedron: Asymmetry - Volume 23, Issue 9, 15 May 2012, Pages 650–654