| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1344738 | 1500369 | 2010 | 4 صفحه PDF | دانلود رایگان |
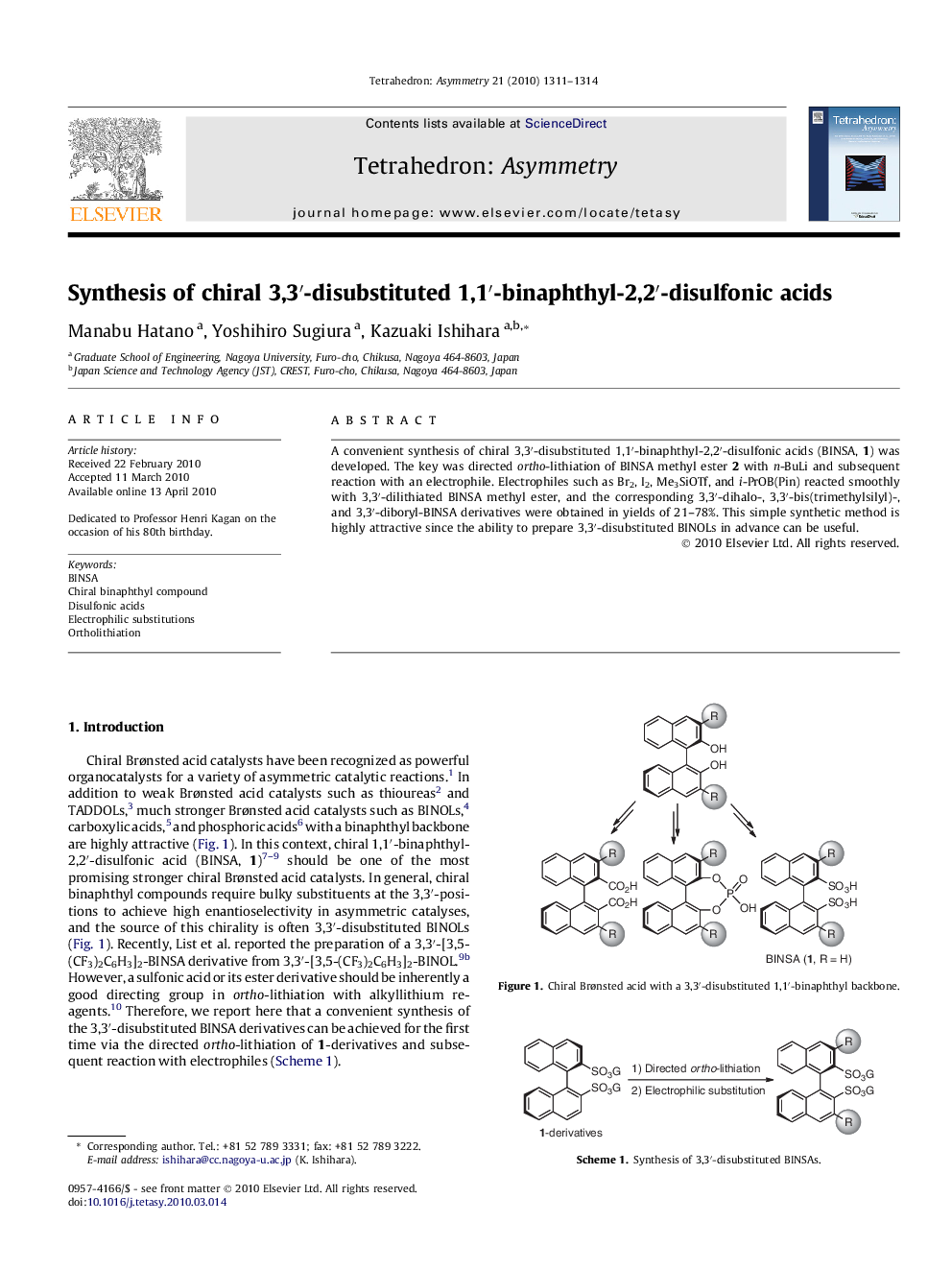
A convenient synthesis of chiral 3,3′-disubstituted 1,1′-binaphthyl-2,2′-disulfonic acids (BINSA, 1) was developed. The key was directed ortho-lithiation of BINSA methyl ester 2 with n-BuLi and subsequent reaction with an electrophile. Electrophiles such as Br2, I2, Me3SiOTf, and i-PrOB(Pin) reacted smoothly with 3,3′-dilithiated BINSA methyl ester, and the corresponding 3,3′-dihalo-, 3,3′-bis(trimethylsilyl)-, and 3,3′-diboryl-BINSA derivatives were obtained in yields of 21–78%. This simple synthetic method is highly attractive since the ability to prepare 3,3′-disubstituted BINOLs in advance can be useful.
Figure optionsDownload as PowerPoint slide
(R)-Dimethyl 1,1′-binaphthalene-2,2′-disulfonateC22H18O6S2[α]D24=+80.8 (c 1.0, CHCl3)Source of chirality: (R)-BINSAAbsolute configuration: (R)
(R)-Diethyl 1,1′-binaphthalene-2,2′-disulfonateC24H22O6S2[α]D24=+31.2 (c 1.0, CHCl3)Source of chirality: (R)-BINSAAbsolute configuration: (R)
(R)-Dimethyl 3,3′-bis(trimethylsilyl)-1,1′-binaphthalene-2,2′-disulfonateC28H34O6S2Si2[α]D26=+77.7 (c 0.17, CHCl3)Source of chirality: (R)-BINSAAbsolute configuration: (R)
Lithium (R)-3,3′-bis(trimethylsilyl)-1,1′-binaphthalene-2,2′-disulfonateC26H28Li2O6S2Si2[α]D28=+95.2 (c 0.29, MeOH)Source of chirality: (R)-BINSAAbsolute configuration: (R)
(R)-Dimethyl 3,3′-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1,1′-binaphthalene-2,2′-disulfonateC34H40B2O10S2[α]D28=+96.9 (c 1.85, CHCl3).Source of chirality: (R)-BINSAAbsolute configuration: (R)
(R)-3,3′-Diborono-1,1′-binaphthyl-2,2′-disulfonic acidC20H16B2O10S2[α]D28=+80.0 (c 1.0, MeOH).Source of chirality: (R)-BINSAAbsolute configuration: (R)
Journal: Tetrahedron: Asymmetry - Volume 21, Issues 9–10, 17 May 2010, Pages 1311–1314